Product Images Rosuvastatin Calcium
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 7 images provide visual information about the product associated with Rosuvastatin Calcium NDC 70518-4143 by Remedyrepack Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Remedy_Label - Remedy Label

This is a description for a box of Rosuvastatin tablets containing 30 tablets of 10 mg each. The manufacturer is Glenmark located in Mahwah, NJ. The medication is RX Only with an NDC number of 70518-4143-00. It should be stored at a temperature of 20-25°C (58-77°F) with excursions permitted to 15.30°C (59-86°F). The directions for use can be found in the package insert. Additionally, the medication should be kept out of reach of children.*
MM2 - Rosuvastatin 10mg 70518 4143 01

This is a description of Rosuvastatin, a 10 mg tablet that is available by prescription only. It is manufactured by Glenmark in Mahwah, NJ. The medication must be stored at 20-25°C (58-77°F) with excursions permitted to 15.30°C (59-86°F) as per USP guidelines. The directions for use can be found in the package insert. It is important to keep this medication out of the reach of children. The NDC number is 70518-4143-01 with an expiration date and lot number to be checked.*
MM3 - Rosuvastatin 10mg 70518 4143 02

This is a package label for Rosuvastatin 10 mg tablets, with a quantity of 90 tablets. The medication is manufactured by Glenmark in Mahwah, NJ, and repackaged by Remedy Repack in Indiana, PA. The prescription is only for an RX and the NDC number for the product is 70518-4143-02. The storage instructions advise keeping the medication at 20-25°C (58-77°F) with excursions permitted to 15-30°C (59-86°F). It is also recommended to refer to the package insert for specific directions on usage.*
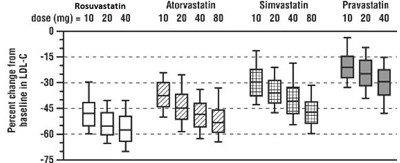
figure1 - figure1
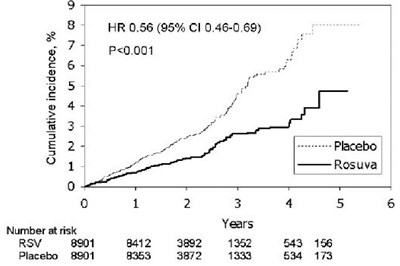
This text provides statistical data on the cumulative incidence of a certain event or phenomenon. It includes information such as the hazard ratio (HR) and confidence interval (CI), as well as p-value indicating statistical significance. The numbers at risk for different groups or conditions are also mentioned.*
figure2 - figure2
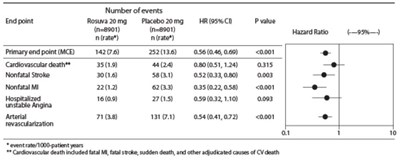
This is a statistical analysis of the efficacy of Rosuvastatin 20 mg compared to Placebo 20 mg in a study with 8901 participants. The data includes various endpoints such as cardiovascular death, nonfatal stroke, nonfatal myocardial infarction (MI), unstable angina, and arterial revascularization. The results show significant reductions in the primary endpoint (Major Cardiovascular Events), nonfatal stroke, nonfatal MI, unstable angina, and arterial revascularization when treated with Rosuvastatin compared to Placebo. The analysis indicates that Rosuvastatin may be effective in reducing cardiovascular risk.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.