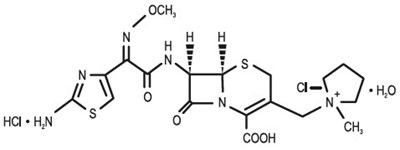
Product Images Cefepime Hydrochloride
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 3 images provide visual information about the product associated with Cefepime Hydrochloride NDC 70594-090 by Xellia Pharmaceuticals Usa Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
PRINCIPAL DISPLAY PANEL - 1 gram Vial Carton - cefepime 02

This is an information leaflet for Cefepime injection. The product is supplied in a sterile, non-pyrogenic, and preservative-free form. It comes in a single 1-gram per vial for intravenous infusion use. The usual dosage and storage instructions are included. Reconstitution instructions are also provided before intramuscular use. The manufacturer is Ol Pharmaceuticals USA, LLC. The NDC number for the drug is 70594-089-02, and it is available in 10 single-dose vials.*
PRINCIPAL DISPLAY PANEL - 2 gram Vial Carton - cefepime 03

Cefepime is a prescription drug used for intravenous infusion. It is available in the form of 10 single-dose vials per package, with each vial containing 2 grams of the drug, and it should be diluted before use. The drug should be stored within the temperature range of 68-77°F or 59-86°F. Additional details such as manufacturer's name and possible side effects are missing in the text.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.