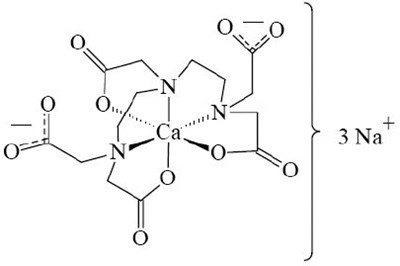
Product Images Pentetate Calcium Trisodium
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 5 images provide visual information about the product associated with Pentetate Calcium Trisodium NDC 70651-001 by Hameln Pharma Gmbh, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
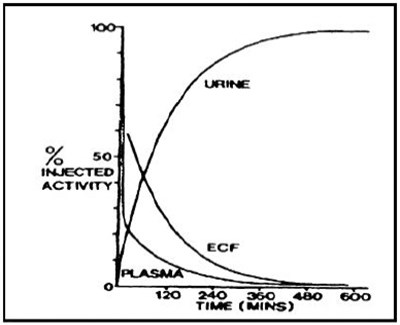
Patient Treatment Data Form - cadtpa 04

This document appears to be a medical report form regarding a patient's treatment data for exposure to radiation or contamination. The form requires information such as the patient's identification, criteria for diagnosis, and initial radioactivity measurement, as well as the route, anatomic area affected, and subsequent decontamination measures taken, such as washing or treatment with Ca-DTPA. The form also contains fields for adverse reactions to treatment and vital signs, as well as disposition and outcome of treatment. A section is also provided for the reporting organization's contact information, and an attachment for emergency records is required.*
PRINCIPAL DISPLAY PANEL - 5 mL Ampoule Package - cadtpa 05
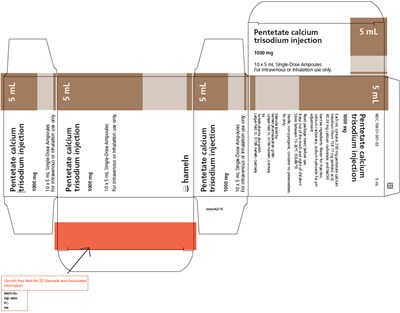
This is a product description of Pentetate calcium trisodium injection in a 105mL single-dose ampoule. It is for intravenous or inhalation use only and is non-pyrogenic. Each container has 1000mg of Pentetate calcium trisodium dissolved in water for injection along with calcium carbonate and sodium hydroxide for pH adjustment. The medicine should be stored between 15-30°C and the package should be read before using it. It is sold under the Mo 70851-001-03 Sm.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.