FDA Label for Nicotine
View Indications, Usage & Precautions
- ACTIVE INGREDIENT STEP ONE (IN EACH PATCH)
- ACTIVE INGREDIENT STEP TWO (IN EACH PATCH)
- ACTIVE INGREDIENT STEP THREE (IN EACH PATCH)
- PURPOSE
- USES
- ASK A DOCTOR BEFORE USE IF YOU HAVE
- ASK A DOCTOR OR PHARMACIST BEFORE USE IF YOU ARE
- WHEN USING THIS PRODUCT
- STOP USE AND ASK A DOCTOR IF
- KEEP OUT OF REACH OF CHILDREN AND PETS
- DIRECTIONS
- OTHER INFORMATION
- INACTIVE INGREDIENTS
- COMMENTS OR QUESTIONS?
- TO INCREASE YOUR SUCCESS IN QUITTING:
- PRINCIPAL DISPLAY PANEL - STEP ONE 21 MG CARTON
- PRINCIPAL DISPLAY PANEL - STEP TWO 14 MG CARTON
- PRINCIPAL DISPLAY PANEL - STEP THREE 7 MG CARTON
Nicotine Product Label
The following document was submitted to the FDA by the labeler of this product Strategic Sourcing Services Llc. The document includes published materials associated whith this product with the essential scientific information about this product as well as other prescribing information. Product labels may durg indications and usage, generic names, contraindications, active ingredients, strength dosage, routes of administration, appearance, warnings, inactive ingredients, etc.
Active Ingredient Step One (In Each Patch)
Nicotine, 21 mg delivered over 24 hours
Active Ingredient Step Two (In Each Patch)
Nicotine, 14 mg delivered over 24 hours
Active Ingredient Step Three (In Each Patch)
Nicotine, 7 mg delivered over 24 hours
Purpose
Stop smoking aid
Uses
Reduces withdrawal symptoms, including nicotine craving, associated with quitting smoking
Ask A Doctor Before Use If You Have
- heart disease, recent heart attack, or irregular heartbeat. Nicotine can increase your heart rate.
- high blood pressure not controlled with medication. Nicotine can increase your blood pressure.
- an allergy to adhesive tape or skin problems because you are more likely to get rashes.
Ask A Doctor Or Pharmacist Before Use If You Are
- using a non-nicotine stop smoking drug
- taking a prescription medicine for depression or asthma. Your prescription dose may need to be adjusted.
When Using This Product
- do not smoke even when not wearing the patch. The nicotine in your skin will still be entering your bloodstream for several hours after you take off the patch.
- if you have vivid dreams or other sleep disturbances, remove this patch at bedtime
Stop Use And Ask A Doctor If
- skin redness caused by the patch does not go away after four days, or your skin swells, or you get a rash
- irregular heartbeat or palpitations occur
- you get symptoms of nicotine overdose, such as nausea, vomiting, dizziness, weakness, and rapid heartbeat
Keep Out Of Reach Of Children And Pets
Used patches have enough nicotine to poison children and pets. If swallowed, get medical help or contact a Poison Control Center right away. Save pouch to use for patch disposal. Dispose of used patch by folding sticky ends together and putting in pouch.
Directions
- if you are under 18 years of age, ask a doctor before use
- before using this product, read the enclosed self-help guide for complete directions and other information
- begin using the patch on your quit day
- if you smoke more than 10 cigarettes per day, use the following schedule below:
- if you smoke 10 or less cigarettes per day, start with Step 2 for 6 weeks, then Step 3 for 2 weeks
- apply one new patch every 24 hours on skin that is dry, clean, and hairless
- remove backing from patch and immediately press onto skin. Hold for 10 seconds.
- wash hands after applying or removing patch. Save pouch to use for patch disposal. Dispose of used patch by folding sticky ends together and putting in pouch.
- the used patch should be removed and a new one applied to a different skin site at the same time each day
- if you have vivid dreams, you may remove the patch at bedtime and apply a new one in the morning
- do not wear more than one patch at a time
- do not cut patch in half or into smaller pieces
- do not leave patch on for more than 24 hours because it may irritate your skin and loses strength after 24 hours
- it is important to complete treatment. If you feel you need to use the patch for a longer period to keep from smoking, talk to your health care provider.
| Weeks 1 thru 4 | Weeks 5 and 6 | Weeks 7 and 8 |
| STEP 1 | STEP 2 | STEP 3 |
| use one 21 mg patch/day | use one 14 mg patch/day | use one 7 mg patch/day |
Other Information
Store at 20°C-25°C (68°F-77°F)
Inactive Ingredients
acrylate adhesive, polyester, silicone adhesive
Comments Or Questions?
1-844-562-7963
To Increase Your Success In Quitting:
1. You must be motivated to quit.
2. Use one patch daily according to directions.
3. It is important to complete treatment.
4. If you feel you need to use the patch for a longer period to keep from smoking, talk to your health care provider.
5. Use patch with a behavioral support program such as the one described in the enclosed booklet.
Principal Display Panel - Step One 21 Mg Carton
Principal Display Panel - Step 1 – 21 mg Carton Label
McKesson SunMark®
NDC 70677-0032-1
COMPARE TO THE ACTIVE INGREDIENT IN HABITROL®*
Clear
Nicotine
Transdermal System
21 mg delivered over 24 hours
STOP SMOKING AID
21 mg
Includes Behavior Support Program with Self-Help Guide
STEP ONE
If you smoke MORE than 10 cigarettes per day: Start with Step 1
If you smoke 10 or LESS cigarettes per day: Start with Step 2
14 Patches
*Habitrol® is a registered trademark of Novartis Consumer Health, Inc.
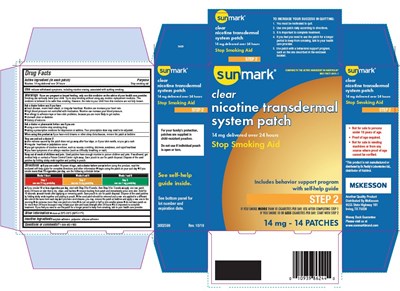
Principal Display Panel - Step Two 14 Mg Carton
Principal Display Panel - Step 2 – 14 mg Carton Label
McKesson SunMark®
NDC 70677-0031-1
COMPARE TO THE ACTIVE INGREDIENT IN HABITROL®*
Clear
Nicotine
Transdermal System
14 mg delivered over 24 hours
STOP SMOKING AID
14 mg
Includes Behavior Support Program with Self-Help Guide
STEP ONE
If you smoke MORE than 10 cigarettes per day: Start with Step 1
If you smoke 10 or LESS cigarettes per day: Start with Step 2
14 Patches
*Habitrol® is a registered trademark of Novartis Consumer Health, Inc.
Principal Display Panel - Step Three 7 Mg Carton
Principal Display Panel - Step 3 – 7 mg Carton Label
McKesson SunMark®
NDC 70677-0030-1
COMPARE TO THE ACTIVE INGREDIENT IN HABITROL®*
Clear
Nicotine
Transdermal System
7 mg delivered over 24 hours
STOP SMOKING AID
7 mg
Includes Behavior Support Program with Self-Help Guide
STEP ONE
If you smoke MORE than 10 cigarettes per day: Start with Step 1
If you smoke 10 or LESS cigarettes per day: Start with Step 2
14 Patches
*Habitrol® is a registered trademark of Novartis Consumer Health, Inc.
* Please review the disclaimer below.