FDA Label for Laxative
View Indications, Usage & Precautions
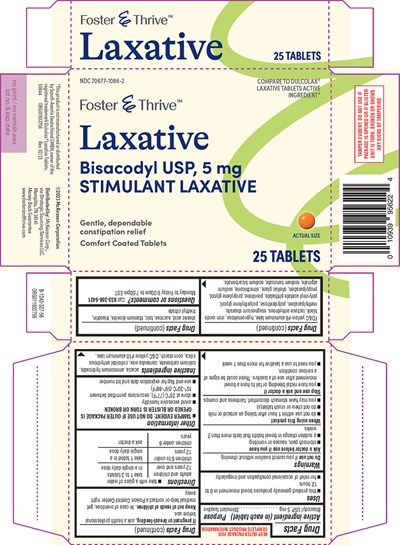
- ACTIVE INGREDIENT (IN EACH TABLET)
- PURPOSE
- USES
- DO NOT USE
- ASK A DOCTOR BEFORE USE IF YOU HAVE
- WHEN USING THIS PRODUCT
- STOP USE AND ASK A DOCTOR IF
- IF PREGNANT OR BREAST-FEEDING,
- KEEP OUT OF REACH OF CHILDREN.
- DIRECTIONS
- OTHER INFORMATION
- INACTIVE INGREDIENTS
- QUESTIONS OR COMMENTS?
- PRINCIPAL DISPLAY PANEL
Laxative Product Label
The following document was submitted to the FDA by the labeler of this product Strategic Sourcing Services Llc. The document includes published materials associated whith this product with the essential scientific information about this product as well as other prescribing information. Product labels may durg indications and usage, generic names, contraindications, active ingredients, strength dosage, routes of administration, appearance, warnings, inactive ingredients, etc.
Active Ingredient (In Each Tablet)
Bisacodyl USP, 5 mg
Purpose
Stimulant laxative
Uses
- this product generally produces bowel movement in 6 to 12 hours
- for relief of occasional constipation and irregularity
Do Not Use
if you cannot swallow without chewing.
Ask A Doctor Before Use If You Have
- stomach pain, nausea or vomiting
- a sudden change in bowel habits that lasts more than 2 weeks
When Using This Product
- do not use within 1 hour after taking an antacid or milk
- do not chew or crush tablet(s)
- you may have stomach discomfort, faintness and cramps
Stop Use And Ask A Doctor If
- you have rectal bleeding or fail to have a bowel movement after use of a laxative. These could be signs of a serious condition.
- you need to use a laxative for more than 1 week
If Pregnant Or Breast-Feeding,
ask a health professional before use.
Keep Out Of Reach Of Children.
In case of overdose, get medical help or contact a Poison Control Center right away.
Directions
- take with a glass of water
| adults and children 12 years and over | take 1 to 3 tablets in a single daily dose |
| children 6 to under 12 years | take 1 tablet in a single daily dose |
| children under 6 years | ask a doctor |
Other Information
- TAMPER EVIDENT: DO NOT USE IF OUTER PACKAGE IS OPENED OR BLISTER IS TORN OR BROKEN
- avoid excessive humidity
- store at 25°C (77°F); excursions permitted between 15°-30°C (59°-86°F)
- see end flap for expiration date and lot number
Inactive Ingredients
acacia, ammonium hydroxide, calcium carbonate, carnauba wax, colloidal anhydrous silica, corn starch, D&C yellow #10 aluminum lake, FD&C yellow #6 aluminum lake, hypromellose, iron oxide black, lactose anhydrous, magnesium stearate, methylparaben, polydextrose, polyethylene glycol, polyvinyl acetate phthalate, povidone, propylene glycol, propylparaben, shellac glaze, simethicone, sodium alginate, sodium benzoate, sodium bicarbonate, stearic acid, sucrose, talc, titanium dioxide, triacetin, triethyl citrate
Questions Or Comments?
Call 833-358-6431 Monday to Friday 9:00am to 7:00pm EST
Principal Display Panel
NDC 70677-1086-2
Foster & Thrive™
COMPARE TO DULCOLAX®
LAXATIVE TABLETS ACTIVE
INGREDIENT*
Laxative
Bisacodyl USP, 5 mg
STIMULANT LAXATIVE
Gentle, dependable
constipation relief
Comfort Coated Tablets
ACTUAL SIZE
25 TABLETS
TAMPER EVIDENT: DO NOT USE IF
PACKAGE IS OPENED OR IF BLISTER
UNIT IS TORN, BROKEN OR SHOWS
ANY SIGNS OF TAMPERING
*This product is not manufactured or distributed
by Sanofi-Aventis Deutschland GMBH, owner of the
registered trademark Dulcolax® Laxative Tablets.
50844 ORG011932756 Rev. 02/23
©2023 McKesson Corporation
Distributed by: McKesson Corp.,
via Strategic Sourcing Services LLC.
Memphis, TN 38141
Money Back Guarantee
www.fosterandthrive.com
* Please review the disclaimer below.