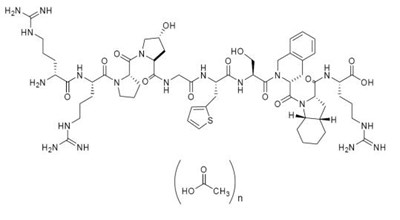
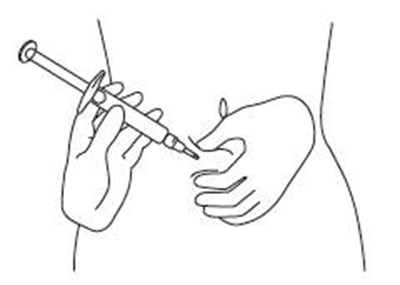
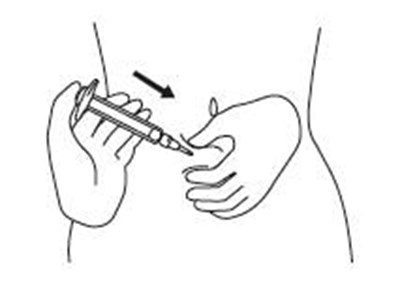
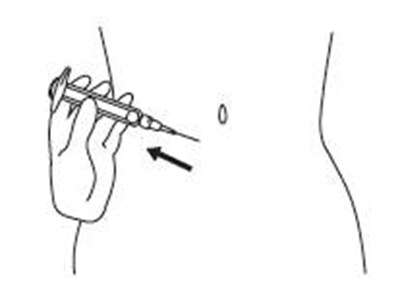
Product Images Sajazir
View Photos of Packaging, Labels & Appearance
- Image - 47e6ba7b 2204 45ec b29f f2baaf56fa65 01
- Image - 47e6ba7b 2204 45ec b29f f2baaf56fa65 02
- Image - 47e6ba7b 2204 45ec b29f f2baaf56fa65 03
- Image - 47e6ba7b 2204 45ec b29f f2baaf56fa65 04
- Image - 47e6ba7b 2204 45ec b29f f2baaf56fa65 05
- Image - 47e6ba7b 2204 45ec b29f f2baaf56fa65 06
- Image - 47e6ba7b 2204 45ec b29f f2baaf56fa65 07
- Image - 47e6ba7b 2204 45ec b29f f2baaf56fa65 08
- Image - 47e6ba7b 2204 45ec b29f f2baaf56fa65 09
- Image - 47e6ba7b 2204 45ec b29f f2baaf56fa65 10
- Image - 47e6ba7b 2204 45ec b29f f2baaf56fa65 11
- Image - 47e6ba7b 2204 45ec b29f f2baaf56fa65 12
- Image - 47e6ba7b 2204 45ec b29f f2baaf56fa65 13
- Image - 47e6ba7b 2204 45ec b29f f2baaf56fa65 14
- Image - 47e6ba7b 2204 45ec b29f f2baaf56fa65 15
- Image - 47e6ba7b 2204 45ec b29f f2baaf56fa65 16
- Image - 47e6ba7b 2204 45ec b29f f2baaf56fa65 17
- 30 of 3 - 47e6ba7b 2204 45ec b29f f2baaf56fa65 18
- 30 mg per 30 mL - 47e6ba7b 2204 45ec b29f f2baaf56fa65 19
- 30mg per 3 lm 1s - 47e6ba7b 2204 45ec b29f f2baaf56fa65 20
- 30mg per 3 mL 3s pack carton - 47e6ba7b 2204 45ec b29f f2baaf56fa65 21
- 30mg per 3 mL 3s pack inner carton - 47e6ba7b 2204 45ec b29f f2baaf56fa65 22
Product Label Images
The following 22 images provide visual information about the product associated with Sajazir NDC 70709-013 by Cycle Pharmaceuticals Ltd-uk, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Image - 47e6ba7b 2204 45ec b29f f2baaf56fa65 03
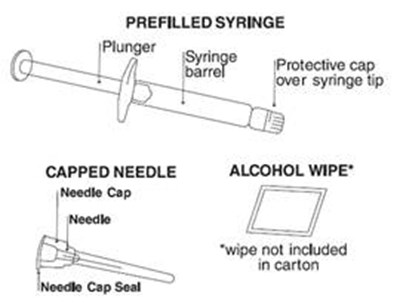
This appears to be a description of a prefilled syringe that includes a plunger and a capped needle, along with an alcohol wipe. Additional information is not available.*
Image - 47e6ba7b 2204 45ec b29f f2baaf56fa65 04

SAJAZIR is a medication that may be administered via injection. The injection contains 30 mg of medication per 3 mL of fluid. The name of the medication appears to be "catibant," although further information is not available.*
30 of 3 - 47e6ba7b 2204 45ec b29f f2baaf56fa65 18

SAJAZIR is a medication administered by subcutaneous injection containing 30 mg/3 mL of icatibant. It comes in a single-use prefilled syringe and is only intended for subcutaneous use. The drug is manufactured by Clpla Ltd. in India at M/s. Gland Pharma Limited, and is distributed by Cycle Pharmaceuticals Ltd. in Cambridge, UK. This information is for prescription use only (Rx Only), and contains an NDC number, a code, and a revision number.*
30 mg per 30 mL - 47e6ba7b 2204 45ec b29f f2baaf56fa65 19

This is a description of a prescription medicine with the trade name SAJAZIR (icatibant) injection manufactured by Cipla Ltd. in India and M/s. Gland Pharma Ltd. in India. It is available in a 3 mL prefilled syringe and is intended for subcutaneous use only. The NDC number for this medicine is 70709-013-03, and it contains 30 mg/3 mL of the active ingredient. The medicine is made for Cycle Pharmaceuticals Ltd. in the UK.*
30mg per 3 lm 1s - 47e6ba7b 2204 45ec b29f f2baaf56fa65 20

SAJAZIR is a prescription drug administered via subcutaneous injection. It is available as a single-dose, single-use, prefilled syringe that contains 30 mg/3 mL (10 mg/mL) of icatibant. The carton includes one syringe and one 25 G hypodermic needle. The syringe comes with a protective cap and should be stored between 2°C - 25°C (36°F - 77°F) and not frozen. SAJAZIR is manufactured by Cipla Ltd., India, and distributed by Cycle Pharmaceuticals Ltd, UK. The drug information, dosage, and administration are provided in the package insert.*
30mg per 3 mL 3s pack carton - 47e6ba7b 2204 45ec b29f f2baaf56fa65 21

This is a product description for SAJAZIR (icatibant) injection, a prescription medication contained in a single-use, single-dose, prefilled syringe for subcutaneous use only. Each syringe contains 30 mg of icatibant in 3 mL and is closed with a protective cap. The inactive ingredients include sodium chloride, glacial acetic acid, sodium hydroxide, and water for injection. The carton also comes with one 26 gauge hypodermic needle and a package insert for full prescribing information, dosage and administration, along with full injection instructions for patients. It is manufactured by Cipla Ltd. in India and is sold and distributed by Cycle Pharmaceuticals Ltd. in Cambridge, UK. The recommended storage is between 2°C - 25°C (36°F - 77°F), without freezing. Batch overprinting occurs in an unvarnished area.*
30mg per 3 mL 3s pack inner carton - 47e6ba7b 2204 45ec b29f f2baaf56fa65 22

This is a description of a medication contained in a prefilled syringe. The medication is Icatibant injection. It comes in a package with three cartons, each containing one single-dose, single-use pre-filled syringe with 25 G hypodermic needles. The prescribing information and patient injection instructions are printed on the package. The syringe contains 30 mg/3 mL of the medication, which is a subcutaneous injection. The medication is for prescription use only and should not be frozen. It is manufactured by Cipla Ltd. in India and by Gland Pharma Limited in India. The product seems to be produced for Cycle Pharmaceuticals Ltd. in Cambridge, UK.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.