Product Images Bortezomib
View Photos of Packaging, Labels & Appearance
- Image - a22e9a33 0c4c 4e0c 8eaf 991e66c50753 01
- Image - a22e9a33 0c4c 4e0c 8eaf 991e66c50753 02
- Image - a22e9a33 0c4c 4e0c 8eaf 991e66c50753 03
- Image - a22e9a33 0c4c 4e0c 8eaf 991e66c50753 04
- Image - a22e9a33 0c4c 4e0c 8eaf 991e66c50753 05
- Image - a22e9a33 0c4c 4e0c 8eaf 991e66c50753 06
- Image - a22e9a33 0c4c 4e0c 8eaf 991e66c50753 07
- Vial Label - a22e9a33 0c4c 4e0c 8eaf 991e66c50753 08
- Carton - a22e9a33 0c4c 4e0c 8eaf 991e66c50753 09
- Carton (Inside flap) - a22e9a33 0c4c 4e0c 8eaf 991e66c50753 10
Product Label Images
The following 10 images provide visual information about the product associated with Bortezomib NDC 70710-1411 by Zydus Pharmaceuticals Usa Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Image - a22e9a33 0c4c 4e0c 8eaf 991e66c50753 06
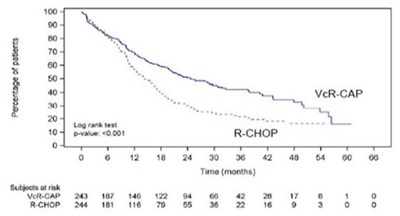
This appears to be a graph showing the number of patients over time and their response to two different treatments, "VCR.CAP" and "RCHOP". The treatments may be related to cancer therapy, as "R-CHOP" is a standard chemotherapy regimen used to treat some lymphomas. However, without additional context, it is not possible to determine the specific conditions or outcomes being measured.*
Image - a22e9a33 0c4c 4e0c 8eaf 991e66c50753 07
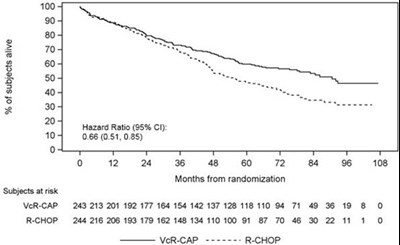
This appears to be a partial result from a clinical trial comparing two treatments, with data on patient survival ("subjects alive"), a hazard ratio, and a chart showing patient outcomes over time for both treatments ("VeR-CAP" and "R-CHOP"). However, without further context or additional information, it is not possible to provide a more specific or useful description.*
Vial Label - a22e9a33 0c4c 4e0c 8eaf 991e66c50753 08

This is a product description for a medication called Bortezomib for Injection, used for intravenous or subcutaneous use. Each vial contains 3.5 mg of bortezomib and 35 mg of mar. The usual dosage varies and should be found on the accompanying packaging. Storage should be at 25°C, and unused portions should be discarded. There are specific excursions permitted for temperature, as specified. Natural materials were not used in the manufacturing of the container closure. The medication is manufactured by Zys Hospira Oncology Pt Ltd in Anmedehad, I, and distributed by Zycus Phamaceuticals (USA) Inc. in Penringon, NJ. Code No: G DRUBSIGIZA26T.*
Carton - a22e9a33 0c4c 4e0c 8eaf 991e66c50753 09
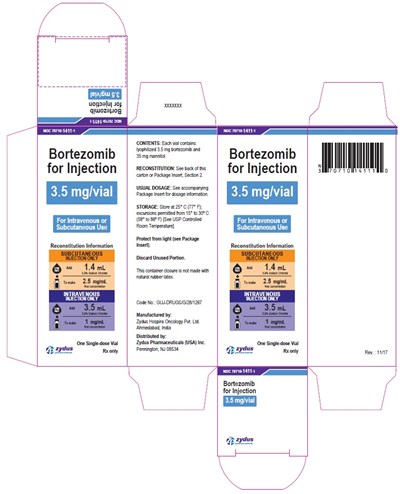
This is a medication instruction for Bortezomib, which comes in the form of an injection for intravenous or subcutaneous use. Each vial contains 3.5 mg/vial of reconstituted Bortezomib. The medication should be stored between 15°C to 30°C and should be protected from light. The manufacturer is Zydus Pharmaceuticals and the medication is only available with a prescription. Details on reconstitution can be found on page 2 of the package insert.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.





