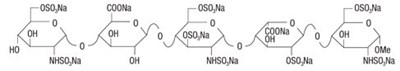
Product Images Fondaparinux Sodium
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 16 images provide visual information about the product associated with Fondaparinux Sodium NDC 70710-1514 by Zydus Pharmaceuticals (usa) Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
1406

This is a product description for Fondaparinux Sodium Injection USP, containing 2.6mg/0.6mL, manufactured by Italfarmaco S.p.A. and distributed by Zydus Pharmaceuticals (USA) Inc. It's subcutaneously used and comes in 10 x 0.5 mL single dose prefilled syringes affixed with automatic needle protection. Fondaparinux Sodium Injection contains 2.5mg/0.5mL isotonic solution of sodium chloride, water for injection, and may contain sodium hydroxide/hydrochloric acid as pH adjusters. It contains dry natural latex rubber in the needle guard that may cause allergic reactions for latex-sensitive individuals. It is recommended to use 2.5mg subcutaneously once daily. This product should be stored between 20°C to 25°C and discarded if unused.*
1409

The text is a product label for Fondaparinux Sodium Injection USP, a medication that is administered by subcutaneous injection. It comes in 2x0.5mL pre-filled syringes, each containing 2.5 mg of fondaparinux sodium in 0.5 mL of an isotonic solution of sodium chloride and water for injection. The prefilled syringe is affixed with an automatic needle protection system. The recommended dose is 2.5 mg subcutaneous injection, once daily. The product is distributed by and manufactured by Zydus Pharmaceuticals (USA) Inc. The package insert should be consulted for additional information.*
1506

This is a description of an injectable medication named "Fondaparinux Sodium Injection USP". It comes in single-dose, prefilled syringes with an automatic needle protection system, and each syringe contains 5mg of fondaparinux sodium. It is intended for subcutaneous use and should only be administered once a day. The description warns of the presence of natural latex rubber in the needle guard of the prefilled syringe, which may cause an allergic reaction in sensitive individuals. The medication should be stored within a specific temperature range and any unused portion should be discarded. There are manufacturer and distributor names on the packaging, as well as a National Drug Code (NDC) and a Global Trade Item Number (GTIN).*
1509

This is a product description for Fondaparinux Sodium Injection USP, a medication used for subcutaneous injection. Each prefilled syringe contains 5 mg of fondaparinux sodium in 0.4 mL of sodium chloride solution. The syringe is prefilled and affixed with an automatic needle protection system. The recommended dose is 5mg once daily. The medication should be stored at 20°C to 25°C (68°F to 77°F) and should be discarded if unused. The package contains two prefilled syringes. The text also includes product information such as the NDC number and the manufacturer's details. The following text is a barcode containing information such as the GTIN, serial number, expiry date, and batch number. The remaining text is a list of numbers that is not comprehensible.*
1606

This is a product label for Fondaparinux Sodium Injection USP, which is intended for subcutaneous use. The injection is available in 10 prefilled syringes of 0.6 mL each and contains 7.5 mg of fondaparinux sodium in an isotonic solution of sodium chloride and water for injection. It may also contain pH adjusters like sodium hydroxide and/or hydrochloric acid. The needle guard of the prefilled syringe contains natural latex rubber, which may cause an allergic reaction in individuals who are sensitive to latex. The recommended dose is 7.5 mg subcutaneous injection once daily. The injection is manufactured by Italfarmaco S.p.A. and distributed by Zydus Pharmaceuticals (USA) Inc. It should be stored at a controlled room temperature of 20°C to 25°C (68°F to 77°F), and unused portions should be discarded.*
1609

This is a description of Fondaparinux Sodium Injection USP, available in single-dose prefilled syringes for subcutaneous use. It contains 7.5 mg of fondaparinux sodium in each 0.6 mL syringe and may also contain sodium hydroxide and/or hydrochloric acid as pH adjusters. The needle guard of the syringe contains dry natural latex rubber that may cause allergic reactions in latex-sensitive individuals. The recommended dose is 7.5 mg subcutaneous injection, once daily. The medication is distributed by Zydus Pharmaceuticals (USA) Inc. and manufactured by Italfarmaco S.p.A. It comes with an automatic needle protection system and instructions to discard unused portions.*
1706

This is a product label for Fondaparinux Sodium Injection used for subcutaneous administration, containing 10 single dose, prefilled syringes each with 10mg /0.8mL of isotonic solution. The product is manufactured by Istituto Lusofarmaco d'Italia S.p.A., and distributed by Zydus Pharmaceuticals (USA) Inc. The product contains natural latex rubber, which may cause allergies in sensitive individuals. It is stored at 20°C to 25°C, and unused portions must be discarded.*
1709

This is a description of Fondaparinux Sodium Injection USP sold in two prefilled syringes. These syringes are meant for subcutaneous use and contain 10mg of fondaparinux sodium in 0.8 ml of isotonic solution that also contains sodium chloride and water for injections. The needle guard in the syringe contains dry natural latex rubber, which may cause allergic reactions in latex-sensitive individuals. The recommended dose is one 10mg subcutaneous injection daily. The drug should be stored at 20°C to 25°C and discarded after use. The product includes a GS1 Datamatrix with a GTIN, a serial number, an expiry date, and a batch number.*
selfinj1 - selfinj1

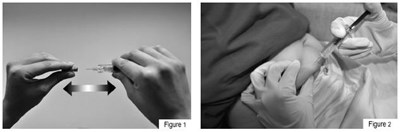
This text provides instructions for self-administration of Fondaparinux sodium injection using a safety syringe consisting of a rigid needle guard, plunger, finger-grip and security sleeve. The text explains how to use the syringe before and after use, as well as how to prepare for the injection by washing hands and choosing a comfortable position. It advises choosing a spot on the lower stomach area, cleaning the injection area with an alcohol swab, and alternating sides for each injection. This text is useful for individuals who need to self-administer Fondaparinux sodium injections.*
selfinj2 - selfinj2
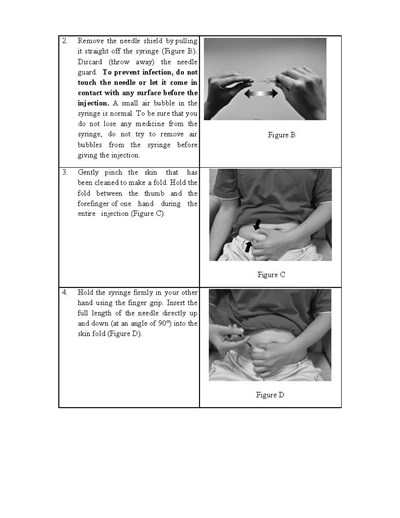
This text provides instructions for safely administering an injection using a syringe. It advises removing the needle shield, pinching the skin to create a fold, and inserting the full length of the needle directly into the fold at a 90-degree angle. The text cautions against touching the needle or letting it come into contact with any surface before injection in order to prevent infection. It also notes that a small air bubble in the syringe is normal and should not be removed in order to not lose any medicine.*
selfinj3 - selfinj3

The text provides instructions on how to use a syringe and activate the safety system to cover the needle after injection to prevent injury. It advises users to inject all the medicine by pressing down on the plunger, remove the needle, and activate the safety system by pushing the plunger rod, making sure to keep the needle away from others. The safety system can be activated only after the syringe is empty and while directing it away from oneself, and activation may cause minimal fluid splatter. The needle shield should not be replaced after injection, and the safety system should not be sterilized.*
selfinj4 - selfinj4

This text provides instructions for proper disposal of used fondaparinux sodium injection needles and syringes. It advises the use of an FDA-cleared sharps disposal container or a household container that is leak-resistant, made of heavy-duty plastic, has a tight-fitting puncture-resistant lid and is properly labeled. Community guidelines must be followed for the right way to dispose of the sharps disposal container. Manufacturing and distribution information of fondaparinux sodium injection is also provided.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.