Product Images Enoxaparin Sodium
View Photos of Packaging, Labels & Appearance
- Figure A - bdf0248f 2aff 42cd 8bf4 639c0ae2d913 01
- Figure B - bdf0248f 2aff 42cd 8bf4 639c0ae2d913 02
- Figure C - bdf0248f 2aff 42cd 8bf4 639c0ae2d913 03
- Figure D - bdf0248f 2aff 42cd 8bf4 639c0ae2d913 04
- Figure E - bdf0248f 2aff 42cd 8bf4 639c0ae2d913 05
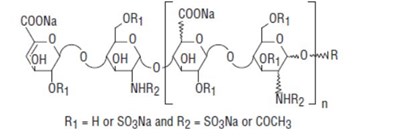
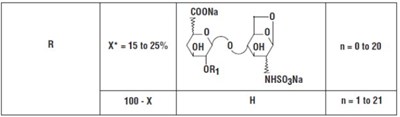
- Structural Formula - bdf0248f 2aff 42cd 8bf4 639c0ae2d913 06
- Structural Formula 2 - bdf0248f 2aff 42cd 8bf4 639c0ae2d913 07
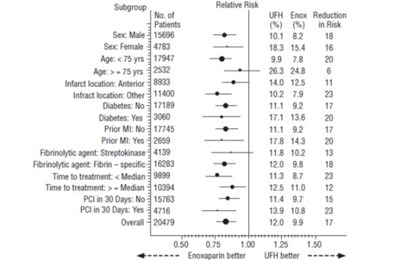
- Figure 1 - bdf0248f 2aff 42cd 8bf4 639c0ae2d913 08
- figure 2 - bdf0248f 2aff 42cd 8bf4 639c0ae2d913 09
- Image - bdf0248f 2aff 42cd 8bf4 639c0ae2d913 10
- Image - bdf0248f 2aff 42cd 8bf4 639c0ae2d913 11
- Image - bdf0248f 2aff 42cd 8bf4 639c0ae2d913 12
- Image - bdf0248f 2aff 42cd 8bf4 639c0ae2d913 13
- Image - bdf0248f 2aff 42cd 8bf4 639c0ae2d913 14
- Image - bdf0248f 2aff 42cd 8bf4 639c0ae2d913 15
- image - bdf0248f 2aff 42cd 8bf4 639c0ae2d913 16
Product Label Images
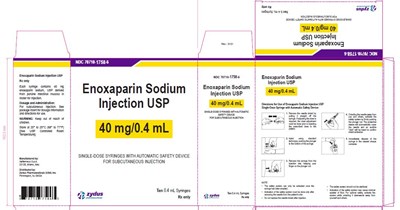
The following 16 images provide visual information about the product associated with Enoxaparin Sodium NDC 70710-1757 by Zydus Pharmaceuticals Usa Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
figure 2 - bdf0248f 2aff 42cd 8bf4 639c0ae2d913 09

This text appears to be a graphical depiction of a distribution function comparison between two treatments (Enoxaparin and UFH) using a Logrank test. The horizontal axis is labeled "Time to Death or Myocardial Reinfarction (days)" and the vertical axis contains the "Number at risk." There is a significant difference between the two treatments, with Enoxaparin being more effective, as indicated by the p-value of less than 0.0001.*
Image - bdf0248f 2aff 42cd 8bf4 639c0ae2d913 10

Enoxaparin Sodium Injection USP is a medication that comes in a package of ten 0.3mL syringes with an automatic safety device for subcutaneous injection. The medication is used in the prevention of deep vein thrombosis and pulmonary embolism. The product has a specific identification number (NDC 7071017575) and is labeled as single-dose.*
Image - bdf0248f 2aff 42cd 8bf4 639c0ae2d913 12

This is a drug label for Enoxaparin Sodium Injection USP, which is a medication administered via subcutaneous injection using single-dose syringes with an automatic safety device. The label includes the National Drug Code (NDC) 70710.17535 and the product code T65LI-0401 0N, along with the quantity of ten 0.6 mL syringes. The label is produced by Zydus pharmaceuticals.*
Image - bdf0248f 2aff 42cd 8bf4 639c0ae2d913 14

This text is a description of the medication Enoxaparin Sodium, in the form of single-dose syringes with an automatic safety device for subcutaneous injection. The package contains ten 1mL syringes, and the concentration of Enoxaparin Sodium is 100mg/mL. Additionally, there are some code and batch numbers visible.*
Image - bdf0248f 2aff 42cd 8bf4 639c0ae2d913 15

This appears to be information related to a medication called "Enoxaparin Sodium Injection USP". It includes details such as the NDC code and product number, as well as information about the syringe used for subcutaneous injection. Not much can be inferred beyond this without further context.*
image - bdf0248f 2aff 42cd 8bf4 639c0ae2d913 16

This appears to be a label or packaging description for a medication called Enoxaparin Sodium Injection USP. It contains important information such as the NDC code, dosage instructions, and the fact that it is packaged in single-dose syringes with an automatic safety device for subcutaneous injection.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.