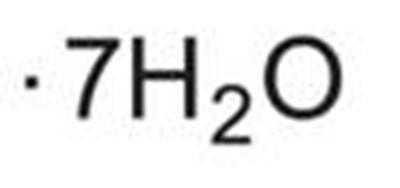Product Images Zinc Sulfate
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 8 images provide visual information about the product associated with Zinc Sulfate NDC 70710-1878 by Zydus Pharmaceuticals Usa Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
10mg container - 7bf7761a 873d 48bc b884 818f16215689 01

This text appears to be a description of a medication called Zinc Sulfate Injection, USP. Each mL of this medication provides 1 mg of zinc. It is intended for intravenous use after dilution and admixing. The medication comes in a 10mL vial and should not be directly infused. It is sterile and contains no more than 1,500 meglL of aluminum. The recommended storage temperature is 20°C to 25°C (68°F to 77°F). Any unused portion should be discarded. The recommended dose should be obtained from the Prescribing Information. The medication is manufactured by Zydus Lifesciences Limited in Vadodara, India and distributed by Zydus Pharmaceuticals (USA) Inc. in Pennington, NJ 08534.*
5mg-container - 7bf7761a 873d 48bc b884 818f16215689 05

This is a description of a zinc sulfate injection solution. Each milliliter of the solution provides 5 mg of zinc in the form of zinc sulfate. The pH of the solution is adjusted with sulfuric acid. It contains no more than 2,500 meg/L of aluminum. The solution should be stored at a temperature between 20°C to 25°C (68°F to 77°F). The recommended dose is mentioned in the prescribing information. This product is distributed by Zydus Pharmaceuticals (USA) Inc., located in Pennington, NJ. The solution comes in a sterile 5mL vial and is for injection use only.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.