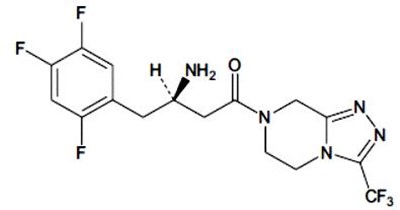
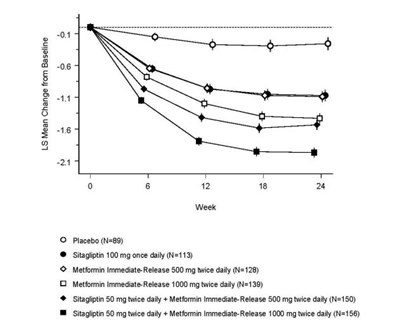
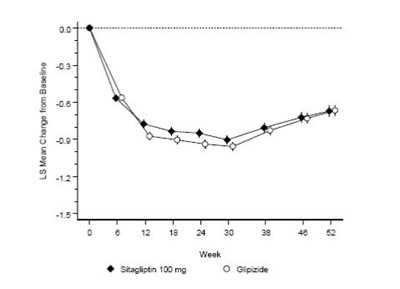
Product Images Sitagliptin And Metformin Hydrochloride
View Photos of Packaging, Labels & Appearance
- Image - 33ea875e cb1b 4508 b538 07828d828e10 01
- Image - 33ea875e cb1b 4508 b538 07828d828e10 02
- Image - 33ea875e cb1b 4508 b538 07828d828e10 03
- Image - 33ea875e cb1b 4508 b538 07828d828e10 04
- 50 mg/ 500 mg - 33ea875e cb1b 4508 b538 07828d828e10 05
- 50 mg/1000 mg - 33ea875e cb1b 4508 b538 07828d828e10 06
- 100 mg/1000 mg - 33ea875e cb1b 4508 b538 07828d828e10 07
Product Label Images
The following 7 images provide visual information about the product associated with Sitagliptin And Metformin Hydrochloride NDC 70710-2038 by Zydus Pharmaceuticals Usa Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
50 mg/ 500 mg - 33ea875e cb1b 4508 b538 07828d828e10 05

Sitagliptin and Metformin Hydrochloride is an oral medication available in 50mg/500mg extended-release tablets. The recommended dosage is two tablets taken once daily with a meal, preferably in the evening. It is important to use the medication within 30 days of opening. The package is child-resistant and should be stored at 20°C to 25°C (68°F to 77°F), with excursions permitted to 15°C to 30°C (59°F to 86°F). Keep the tablets in a dry place with the cap tightly closed and in the original container to protect them from moisture. Contact the provided number for further information or discard the medication after use. Manufactured by Zydus Lifesciences Ltd. in Ahmedabad, India, and distributed by Zydus Pharmaceuticals (USA) Inc. in Pennington, NJ.*
50 mg/1000 mg - 33ea875e cb1b 4508 b538 07828d828e10 06

This is a description of Sitagliptin and Metformin Hydrochloride Extended-Release Tablets, for oral use. Each film-coated tablet contains Sitagliptin 50 mg and Metformin Hydrochloride, USP 1,000 mg. The recommended dosage is two tablets taken once daily with a meal, preferably in the evening. The package is child-resistant and should be stored at 20°C to 25°C (66°F to 77°F), with excursions permitted from 15°C to 30°C (59°F to 86°F). It should be stored in a dry place with the cap tightly closed and dispensed in the original container. It is advised to keep the tablets in the original container to protect them from moisture. The medication guide is available at zydususa.com/medguides or by calling 1-877-993-8775. This medication is manufactured by Zydus Lifesciences Ltd. in Ahmedabad, India, and distributed by Zydus Pharmaceuticals (USA) Inc. in Pennington, NJ.*
100 mg/1000 mg - 33ea875e cb1b 4508 b538 07828d828e10 07

This text provides information about Sitagliptin and Metformin Hydrochloride Extended-Release Tablets, which contain 100mg of Sitagliptin and 1,000mg of Metformin Hydrochloride each. The tablets are recommended to be taken once daily with a meal, preferably in the evening. The package is child-resistant and should be stored at 20°C to 25°C. The tablets should be used within 30 days of opening and kept in the original container to protect them from moisture. For further guidance or inquiries, contact Zydus Lifesciences Ltd. in Ahmedabad, India, or Zydus Pharmaceuticals (USA) Inc. in Pennington, New Jersey.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.