Product Images Arformoterol Tartrate
View Photos of Packaging, Labels & Appearance
- Image - 9e59e677 5b11 43bf 947a c4d5d8c9b710 01
- Image - 9e59e677 5b11 43bf 947a c4d5d8c9b710 02
- Image - 9e59e677 5b11 43bf 947a c4d5d8c9b710 03
- Image - 9e59e677 5b11 43bf 947a c4d5d8c9b710 04
- Image - 9e59e677 5b11 43bf 947a c4d5d8c9b710 05
- Image - 9e59e677 5b11 43bf 947a c4d5d8c9b710 06
- figure 4 - 9e59e677 5b11 43bf 947a c4d5d8c9b710 07
- Image - 9e59e677 5b11 43bf 947a c4d5d8c9b710 08
- Image - 9e59e677 5b11 43bf 947a c4d5d8c9b710 09
- pouch - 9e59e677 5b11 43bf 947a c4d5d8c9b710 10
- carton - 9e59e677 5b11 43bf 947a c4d5d8c9b710 11
Product Label Images
The following 11 images provide visual information about the product associated with Arformoterol Tartrate NDC 70748-175 by Lupin Pharmaceuticals, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Image - 9e59e677 5b11 43bf 947a c4d5d8c9b710 02
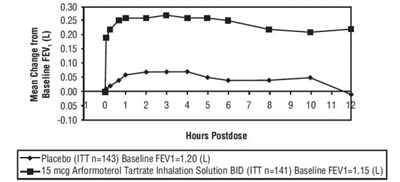
This text appears to be a table with numerical values indicating mean change from basal FEV at various hours postdose for a placebo group and a group treated with 15 mcg Arformoterol Tartrate Inhalation Solution BID. The baseline FEV1 for the placebo group is 1.20 (L) and for the treated group is 1.15 (L). No further context or information is provided.*
pouch - 9e59e677 5b11 43bf 947a c4d5d8c9b710 10

This is a description of a medication called Arformoterol Tartrate that comes in a unit-dose vial. It is an inhalation solution with a potency expressed as arformoterol. The vial contains 15 mg of arformoterol squalene, which equates to 22 mcg of arformoterol. The solution is packed in a pouch and is meant to be opened just before use. The vial should be kept out of reach of children and protected from excessive heat and light. The unopened pouches with unit-dose vials can be stored in a refrigerator at 36°F to 46°F or at a temperature of 20°C to 25°C for up to ten weeks. The medication label includes the lot number and manufacturing details. The product should be used before the expiration date.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.








