Product Images Formoterol Fumarate
View Photos of Packaging, Labels & Appearance
- Image - aad1bcb4 df57 4e80 9129 7973ebb9dea5 01
- Image - aad1bcb4 df57 4e80 9129 7973ebb9dea5 02
- Image - aad1bcb4 df57 4e80 9129 7973ebb9dea5 03
- Image - aad1bcb4 df57 4e80 9129 7973ebb9dea5 04
- Image - aad1bcb4 df57 4e80 9129 7973ebb9dea5 05
- Image - aad1bcb4 df57 4e80 9129 7973ebb9dea5 06
- Pouch Label - aad1bcb4 df57 4e80 9129 7973ebb9dea5 07
- Carton Label - 30's Pack: - aad1bcb4 df57 4e80 9129 7973ebb9dea5 08
- Carton 60s pack - aad1bcb4 df57 4e80 9129 7973ebb9dea5 09
Product Label Images
The following 9 images provide visual information about the product associated with Formoterol Fumarate NDC 70748-261 by Lupin Pharmaceuticals, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Image - aad1bcb4 df57 4e80 9129 7973ebb9dea5 02

This text shows data on Mean FEV in Liters for patients who used Formoterol fumarate inhalation solution 20 mcg (N=123) versus those who used a placebo (N=114).*
Image - aad1bcb4 df57 4e80 9129 7973ebb9dea5 03
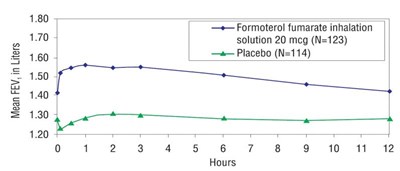
This appears to be a table or graph displaying the mean Forced Expiratory Volume (FEV) in liters for two groups of participants (one receiving Formoterol fumarate inhalation solution 20 mcg and another receiving a placebo), with the x-axis representing the different liters values (ranging from 1.30 to 1.80).*
Pouch Label - aad1bcb4 df57 4e80 9129 7973ebb9dea5 07
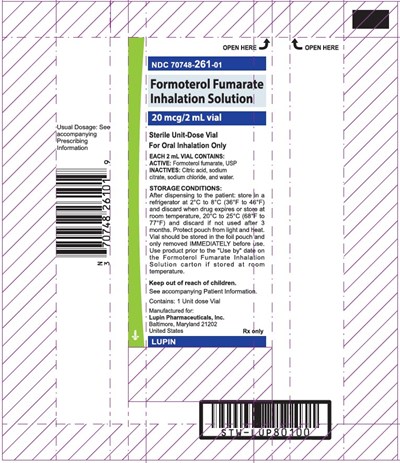
Formoterol Fumarate Inhalation Solution is a medication that comes in 20mcg strength per 2mL vial with a usual dosage of S0. It should only be used for oral inhalation and stored in the refrigerator at 2°C to 8°C (36°F to 46°F) and discarded if not used after 3 months. The vial should be kept in the pouch and only removed immediately before use. The medication should be used prior to the "Use by" date. The product is manufactured by Lupin Pharmaceuticals and contains 1 unit-dose vial. The accompanying patient information should be read prior to use. Keep out of reach of children.*
Carton 60s pack - aad1bcb4 df57 4e80 9129 7973ebb9dea5 09

This is a description of Formoterol Fumarate Inhalation Solution. It provides instructions for storage conditions and usage for healthcare providers and patients. The solution comes in Strle Unit Dose Vials, each of which is individually wrapped. The active ingredient in the vial is Formoterol fumarate, USP, and the vial also contains inactive ingredients. The carton contains 60 individual wrapped 2 mL unit dose vials. The vial should always be stored in the foil pouch, and before dispensing to the patient, it should be stored in a refrigerator. After dispensing to the patient, it should be stored in a refrigerator between 2°C to 8°C (36'F to 46°F), protected from ght and heat and discarded when the drug expires or stored at room temperature between 20°C to 25°C (68°F to 77°F) and not used after three months. Protect the pouch from ght and heat. The solution is Rxonly.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.




