Product Images Arformoterol Tartrate
View Photos of Packaging, Labels & Appearance
- Image - 2d707787 edf5 4b8c ba80 e83c87d47708 01
- Image - 2d707787 edf5 4b8c ba80 e83c87d47708 02
- Image - 2d707787 edf5 4b8c ba80 e83c87d47708 03
- Image - 2d707787 edf5 4b8c ba80 e83c87d47708 04
- Image - 2d707787 edf5 4b8c ba80 e83c87d47708 05
- Image - 2d707787 edf5 4b8c ba80 e83c87d47708 06
- Image - 2d707787 edf5 4b8c ba80 e83c87d47708 07
- Image - 2d707787 edf5 4b8c ba80 e83c87d47708 08
- pouch 1 unit - 2d707787 edf5 4b8c ba80 e83c87d47708 09
- 5s foil - 2d707787 edf5 4b8c ba80 e83c87d47708 10
- 30 carton - 2d707787 edf5 4b8c ba80 e83c87d47708 11
- 60 - 2d707787 edf5 4b8c ba80 e83c87d47708 12
Product Label Images
The following 12 images provide visual information about the product associated with Arformoterol Tartrate NDC 70756-612 by Lifestar Pharma Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Image - 2d707787 edf5 4b8c ba80 e83c87d47708 02
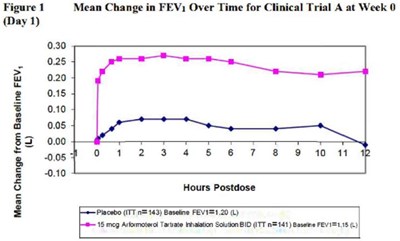
The text describes a figure (Figure 1) showing a comparison of mean change in FEV (Forced Expiratory Volume) from baseline between two groups over Hours Postdose. One group received placebo (n=143) and had a baseline FEV1 of 1.20 L, while the other group received 15 mcg Arformoterol Tartrate inhalation solution BID (n=141) and had a baseline FEV1 of 1.15 L.*
Image - 2d707787 edf5 4b8c ba80 e83c87d47708 03
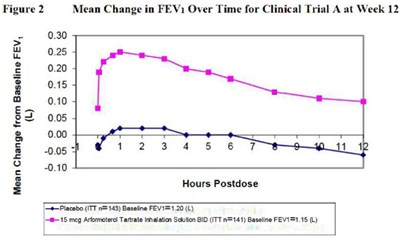
This text describes a figure showing the mean change in Forced Expiratory Volume in 1 second (FEV1) over time for Clinical Trial A at Week 12. The figure shows the changes in FEV1 in Liters (L) for a placebo group (n=143) and a group given Arformoterol Tartrate Inhalation Solution BID (n=141) at baseline FEV1 levels of 1.20 L and 1.15 L, respectively. The data is plotted against hours postdose and the mean change in FEV1 is noted for different time intervals.*
pouch 1 unit - 2d707787 edf5 4b8c ba80 e83c87d47708 09

This is an inhalation solution that is contained in a 2 mL sterile unit-dose vial for Rx use only. The temperature storage and use by date are not available. The solution contains arformoterol tartrate expressed as 15 mcg/2 mL potency, and it should be opened just prior to administration. The text also warns to keep the solution out of children's reach. The rest of the text seems to be not available as it contains non-English characters and errors.*
60 - 2d707787 edf5 4b8c ba80 e83c87d47708 12

Arformoterol Tartrate Inhalation Solution is a bronchodilator medication used for the treatment of breathing problems in patients suffering from chronic obstructive pulmonary disease (COPD). The solution comes in a carton of sixty 2 mL unit-dose vials that need to be refrigerated or stored at room temperature. Each 2 mL unit-dose vial contains 15 μg of arformoterol tartrate inhalation solution, equivalent to 22 μg of arformoterol tartrate in 3 mL of sodium chloride solution. The medication is only available with a prescription, and the proper dosage must be determined by a qualified physician. The package comes with a warning label and should be kept out of children's reach.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.







