Product Images Haloperidol Decanoate
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 16 images provide visual information about the product associated with Haloperidol Decanoate NDC 70756-624 by Lifestar Pharma Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
50mg1mlvial - 74a4c0d9 515d 4e88 bae5 045ea55940ae 02

Each mL of this product, identified by NDC 70756-615-81, contains 50 mg of the medication haloperidol. It should be stored within a temperature range of 20°C to 25°C (68°F to 77°F) and should be protected from light. Excursions from this temperature range are permitted, within limits and should be kept within 15°C to 30°C (59°F to 86°F). The injection is not intended for refrigeration or freezing and is manufactured by Lifestar Pharma LLC in India. This product is provided as a single-dose vial, intended for intramuscular use only. However, due to the poor quality of the text obtained, some information could be inaccurate.*
50mg1mlmonocarton - 74a4c0d9 515d 4e88 bae5 045ea55940ae 03
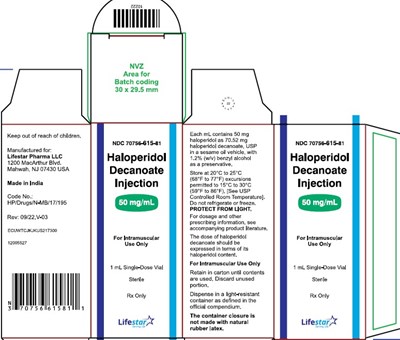
This is a description of a medication for intramuscular use only. It comes in a single-dose vial, and should be kept in a light-resistant container. The dose of the medication is dependent on its haloperidol content. The container closure does not contain natural rubber latex, and must be retained in its carton until use and any unused portion must be discarded. The label includes batch coding and the manufacturer's information.*
50mg1ml3carton - 74a4c0d9 515d 4e88 bae5 045ea55940ae 04
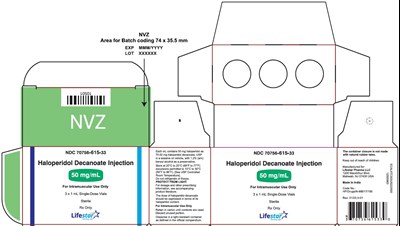
This is a product label for Haloperidol Decanoate injection with dosage information and instructions for use. The injection is for intramuscular use only, sterile, and comes in 3 single-dose vials. Each vial contains 50mg of Haloperidol. The label warns against exposure to light, refrigeration, and emphasizes that the container closure is not made of natural rubber latex. The label contains information related to batch coding, storage, and disposal of the contents.*
50mg1ml10carton - 74a4c0d9 515d 4e88 bae5 045ea55940ae 05

This is a medical label with information on haloperidol decanoate injection. The medication is available in 50mg/mL, to be used intramuscularly only. It comes in a box of 10 single-dose vials, which need to be kept at a controlled room temperature between 20°C to 25°C (68°F to 77°F), protected from light. The package contains details on the Lot, NDC number, and expiry date, which is typically expressed in MMM/YYYY format. The box also includes information on the dosage and pharmacological properties. The label reminds the user to keep the medicine out of children's reach.*
250mg5mlvial - 74a4c0d9 515d 4e88 bae5 045ea55940ae 06

NDC 70756-624-85 is the label of a haloperidol decanoate injection, available in 250 mg/5 with 50 mg/mL concentration. This is a multi-dose vial that is meant to be used intramuscularly. The vial consists of a sterile solution, and storage temperature ranges from 20°C to 25°C (68°F to 77°F), and excursions allowed up to 15°C to 30°C (59°F to 86°F). It is advised not to refrigerate or freeze. It is essential to protect from light. The code on the label is HP/Drugs/N-MB/17/195, and the expiry date indicated is MMM/YYYY, with lot number XXXXXX. Finally, the area in the label is unvarnished and measures 8x20 mm.*
250mg5mlmonocarton - 74a4c0d9 515d 4e88 bae5 045ea55940ae 07
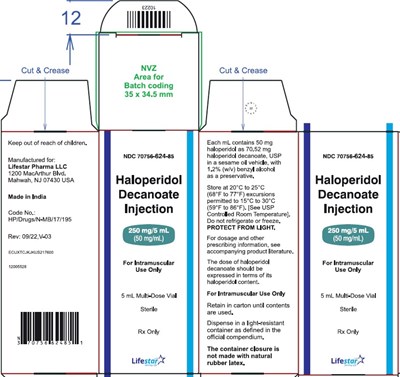
This is a medication label for a 5 mL vial of Haloperidol Decanoate Injection, 50mg/ml, manufactured for Lifestar Pharma LLC. The medication is for intramuscular use only and should be kept at a temperature between 20°C-25°C. It contains haloperidol as haloperidol decanoate, USP in a sesame oil vehicle and has a preservative. The label provides caution statements and dosage information. The dose should be expressed in terms of haloperidol content and the medication should be protected from light.*
250mg5ml10carton - 74a4c0d9 515d 4e88 bae5 045ea55940ae 08

Not available. The text appears to be mostly random characters generated by with minimal actual English words or phrases.*
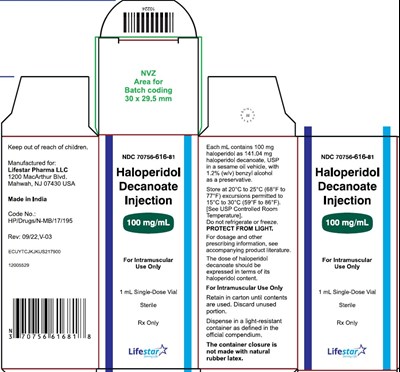
100mg1mlvial - 74a4c0d9 515d 4e88 bae5 045ea55940ae 09

This text describes a medication with NDC code 70756-616-81 that is to be stored at a temperature of 20-25°C (68-77°F) and protected from light. The medication is a haloperidol injection, with each mL containing 100 mg of the active ingredient. The medication is manufactured by Lifestar Pharma LLC in India and is intended only for intramuscular use. The text also provides information on the vial size (1 mL), lot number (XXXXXX), and expiration date (MMM/YYYY), as well as instructions not to refrigerate or freeze the medication.*
100mg1ml - 74a4c0d9 515d 4e88 bae5 045ea55940ae 10
This is a product label for Haloperidol Decanoate Injection, which is intended for intramuscular use only. The manufacturer is Lifestar Pharma LLC, located in Mahwah, NJ, and the product is made in India. Each mL contains 100 mg of haloperidol as 141,04 mg of haloperidol decanoate, USP, in a sesame oil vehicle. The injection is packaged in a 1mL single-dose vial, which is sterile and should be protected from light. The product should be stored at 20°C to 25°C (68°F to 77°F), with excursions permitted to 15°C to 30°C (59°F to 86°F), and should not be refrigerated or frozen. The container closure is not made with natural rubber latex. The label includes important information on proper dosing and prescribing.*
100mg1mlmonocarton - 74a4c0d9 515d 4e88 bae5 045ea55940ae 11
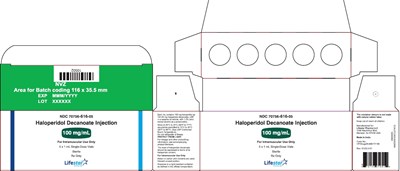
This is a label for Haloperidol Decanoate Injection. It has a batch code area and a section for expiration date, lot number, and NDC number. The medicine is for Intramuscular Use Only, and each 1 mL single-dose vial contains 100 mg/mg of Haloperidol Decanoate. The label has some gibberish characters and the phrase "Proteot o G Frdosagasnd oo sy."*
100mg1ml10carton - 74a4c0d9 515d 4e88 bae5 045ea55940ae 12

This is a product label for Haloperidol Decanoate Injection, indicating its contents, dosage, and administration. The injection is for intramuscular use only and is sterile. The label contains information on how to maintain the product, including temperature storage, light exposure, and disposal of unused portions. The product is not made with natural rubber latex. It is manufactured for Lifestar Pharma LLC, located in New Jersey, USA.*
500mg5mlvial - 74a4c0d9 515d 4e88 bae5 045ea55940ae 13

This is a description of Haloperidol Decanoate Injection, a medication that is administered intramuscularly using a 5 mL multi-dose vial. Each mL contains 100 mg haloperidol and the vial contains 500 mg/5 L. This medication should be stored at 20°C to 25°C (68°F to 77°F) with excursions permitted to 15°C to 30°C (59°F to 86°F). It should be protected from light and should not be refrigerated or frozen. The manufacturer is Lifestar Pharma LLC, located in India. The text also includes batch and expiry date information.*
500mg5ml5carton - 74a4c0d9 515d 4e88 bae5 045ea55940ae 15
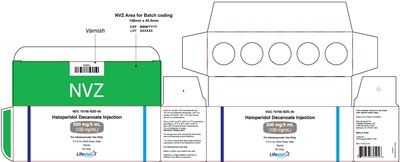
This is a label for an injection of Haloperidol Decanoate. It includes information such as the size of the NVZ area for batch coding, the expiration date, lot number, and dosage information. Additionally, it indicates that this injection is for intramuscular use only, has a sterility guarantee, and is available in a 5 x 5 mL multi-dose vials. The label also includes warnings and instructions for use.*
500mg5ml10carton - 74a4c0d9 515d 4e88 bae5 045ea55940ae 16

This is a description of a Haloperidol Decanoate injection with a concentration of 500mg/5mL (100mg/mL) for intramuscular use only. The injection is contained in a 105mL multi-dose vial. The dose of Haloperidol Decanoate should be expressed in terms of 3 Nefoperidol Cere. The container is sterile and should be kept at a temperature between 20°C and 25°C (68° and 77°F), with excursions permitted to 15° to 30°C (59° to 86°F). The container closure is not made with natural rubber, and it should be kept out of reach of children. The manufacturer is Lifestar Pharma LLC, and the product is made in India. For more information on dosage and prescribing recommendations, refer to the accompanying literature.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.

