Product Images Glycopyrrolate
View Photos of Packaging, Labels & Appearance
- 1 mL vial - 81672c53 d609 4d33 81c2 4d61e6fe09de 01
- carton of 25 of 1 mL vial - 81672c53 d609 4d33 81c2 4d61e6fe09de 02
- 2 mL vial - 81672c53 d609 4d33 81c2 4d61e6fe09de 03
- carton of 2 mL 25 vials - 81672c53 d609 4d33 81c2 4d61e6fe09de 04
- 5 mL vial - 81672c53 d609 4d33 81c2 4d61e6fe09de 05
- carton of 25 of 5 mL vial - 81672c53 d609 4d33 81c2 4d61e6fe09de 06
- 20 mL vial - 81672c53 d609 4d33 81c2 4d61e6fe09de 07
- carton of 10 - 20 mL vials - 81672c53 d609 4d33 81c2 4d61e6fe09de 08
Product Label Images
The following 8 images provide visual information about the product associated with Glycopyrrolate NDC 70771-1619 by Zydus Lifesciences Limited, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
1 mL vial - 81672c53 d609 4d33 81c2 4d61e6fe09de 01

This is a description of a medication called Glycopyrrolate Injection. It is not suitable for use in newborns and should be stored at a temperature between 20 to 25°C. It contains Benzyl Alcohol and should be given either intra-huscularly or intravenously. The medication comes in a vial made in India by GUJDRUGS/GR8/1609. It requires a prescription (Rx only).*
2 mL vial - 81672c53 d609 4d33 81c2 4d61e6fe09de 03

This text provides information about the storage, dosage, and administration details of Glycopyrrolate Injection, a medication used in pharmaceuticals. It is not suitable for use in newborns and contains benzyl alcohol. It can be administered through intramuscular or intravenous means, and it comes in a single dose vial. It is stored at a controlled room temperature ranging from 20 to 25°C.*
5 mL vial - 81672c53 d609 4d33 81c2 4d61e6fe09de 05

This is a medication label containing information on dosage, storage, and ingredients for an injection, which contains glycopyrrolate and benzyl alcohol as a preservative. It is manufactured in India and should not be used in newborns. It is intended for intramuscular or intravenous administration and comes in a 5 mL single-dose vial.*
20 mL vial - 81672c53 d609 4d33 81c2 4d61e6fe09de 07

This is a description of an injectable solution containing water for injection, USP, with a preservative called Benzyl alcohol, NF . It also contains Glycopyrrolate with hydrochloric acid and/or sodium hydroxide. The recommended dosage is 4mg/20mL. The solution should be stored at a temperature between 20 to 25°C (68° to 77°F). It should be noted that the solution contains Benzyl alcohol and is for intramuscular or intravenous administration only. It is a Zydus Pharmaceuticals product and comes in a 20 mL Multiple-Dose Vial.*
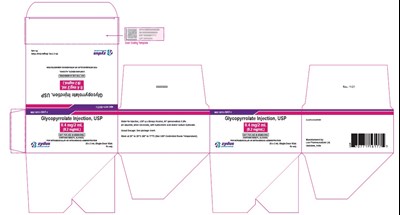
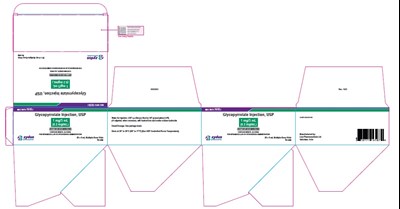
carton of 10 - 20 mL vials - 81672c53 d609 4d33 81c2 4d61e6fe09de 08

This text appears to be a medication label for Glycopyrrolate Injection, USP. It comes in a 20mL vial with a concentration of 0.2mg/mL. It is not clear what "Py s" at the end of the text means in this context.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.