Product Images Oxcarbazepine
View Photos of Packaging, Labels & Appearance
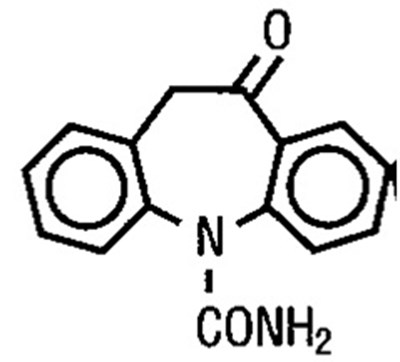
- structure - 6a50f32f 6dab 4f67 b7ac 754cceea4299 01
- figure - 6a50f32f 6dab 4f67 b7ac 754cceea4299 02
- figure - 6a50f32f 6dab 4f67 b7ac 754cceea4299 03
- figure - 6a50f32f 6dab 4f67 b7ac 754cceea4299 04
- figure - 6a50f32f 6dab 4f67 b7ac 754cceea4299 05
- 150mg container label - 6a50f32f 6dab 4f67 b7ac 754cceea4299 06
- 150mg carton label - 6a50f32f 6dab 4f67 b7ac 754cceea4299 07
- 300mg container label - 6a50f32f 6dab 4f67 b7ac 754cceea4299 08
- 300mg carton label - 6a50f32f 6dab 4f67 b7ac 754cceea4299 09
- 600mg container label - 6a50f32f 6dab 4f67 b7ac 754cceea4299 10
- 600mg carton label - 6a50f32f 6dab 4f67 b7ac 754cceea4299 11
Product Label Images
The following 11 images provide visual information about the product associated with Oxcarbazepine NDC 70771-1844 by Zydus Lifesciences Limited, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
figure - 6a50f32f 6dab 4f67 b7ac 754cceea4299 02

The text provides the results of a log-rank test, where the p-value was found to be 0.0001. The data also includes a graphic representation showing Time on trial from Day 2 (days) for the Treatment Group involving Oxcarbazeping.*
figure - 6a50f32f 6dab 4f67 b7ac 754cceea4299 03

This description provides the result of a log-rank test with a p-value of 0.046. The data shows the days from the 1st dose to the 15th seizure for two treatment groups: Oxcarbazepine and Placebo.*
figure - 6a50f32f 6dab 4f67 b7ac 754cceea4299 05

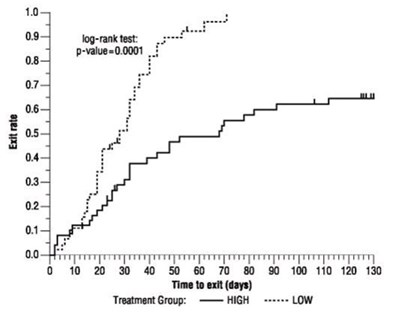
The log-rank test was performed with a resulting p-value of 0.0001. Additionally, there is a series of values from 10 to 130 displayed in the text.*
150mg container label - 6a50f32f 6dab 4f67 b7ac 754cceea4299 06

This text appears to contain pharmaceutical information about Oxcarbazepine USP 150 mg tablets including dosage instructions and storage conditions. It also mentions the manufacturing company - Cadila Healthcare Ltd., located in Baddi, India. Additionally, it provides guidelines for pharmacists to dispense the medication safely.*
150mg carton label - 6a50f32f 6dab 4f67 b7ac 754cceea4299 07

This text appears to be describing Oxcarbazepine Tablets, USP, including information on dosage, storage, and safety considerations. The tablets are supplied in containers and the usual dosage instructions are provided on the package. The text also mentions the manufacturing company and advises to keep the tablets and all drugs out of the reach of children. It is important to follow the instructions provided when using this medication.*
300mg container label - 6a50f32f 6dab 4f67 b7ac 754cceea4299 08

The text provides information about a pharmaceutical product, specifically Oxcarbazepine Tablets, USP, containing 300 mg of Oxcarbazepine. The dosing information, storage guidelines, and manufacturer details are included. It also mentions the need to dispense a Medication Guide to each patient and emphasizes keeping all drugs out of the reach of children. The product is distributed by Zydus Pharmaceuticals and manufactured by Cadila Healthcare Ltd. in Baddi, India.*
300mg carton label - 6a50f32f 6dab 4f67 b7ac 754cceea4299 09

Product Description: Oxcarbazepine Tablets, USP in 300 mg strength. Each tablet contains Oxcarbazepine. The usual dosage information can be found on the package insert for complete prescribing details. This medication should be stored at controlled room temperature. Keep out of reach of children. Available in packs of 100 tablets (in 10x10 unit-dose packaging). Made by Zydus Cadila.*
600mg container label - 6a50f32f 6dab 4f67 b7ac 754cceea4299 10

A description of a medication packaging for Oxcarbazepine Tablets, USP. The packaging includes information on tablet dosage, storage instructions, manufacturer details, and a reminder to dispense the Medication Guide to each patient. It also emphasizes the importance of keeping the medication out of reach of children. Additionally, it mentions that the FDA-approved organic impurities acceptance criteria differs from the USP test. The tablets contain 600 mg of Oxcarbazepine USP.*
600mg carton label - 6a50f32f 6dab 4f67 b7ac 754cceea4299 11

This is a description of Oxcarbazepine Tablets, USP 600 mg in a pack of 100 tablets (10 x 10 unit dose). The tablets are manufactured by Codia tesncar Inc. It includes information such as the active ingredient, usual dosage instructions, storage guidelines, and a reminder to keep all drugs out of the reach of children. The document also mentions that the tablets are dispensed in a container as defined by the Fratic and are compliant with USP standards.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.