Product Images Valsartan And Hydrochlorothiazide
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 5 images provide visual information about the product associated with Valsartan And Hydrochlorothiazide NDC 71205-015 by Proficient Rx Lp, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
71205-015-90 - valsahctz 320 25mg

This is a medication with the NDC code 71205-015-90 containing 320 mg of Valsartan and 25 mg of Hydrochlorothiazide USP. The medication is in the form of yellow, oval-shaped film-coated tablets with 'L239' on one side and plain on the other side. It is produced by Alembic Pharmaceuticals Limited and contains 90 tablets in one package. The medication is labeled and stored properly, and is available in different quantities (30 tablets, and 90 tablets). Proficient Rx LP is the company responsible for relabeling the medication.*
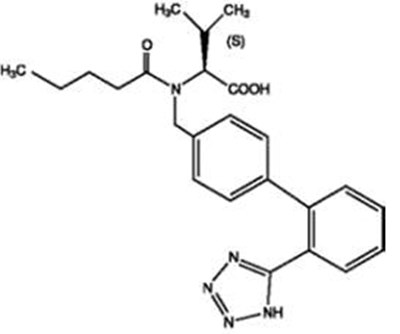
Figure 1 - valsahctz figur1

The text describes a figure showing the probability of achieving systolic blood pressure lower than 140 mmHg at week 8, with different baseline measurements. There are measurements and names of different medicines but the text does not provide more information.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.