Product Images Losartan Potassium
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 6 images provide visual information about the product associated with Losartan Potassium NDC 71205-194 by Proficient Rx Lp, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
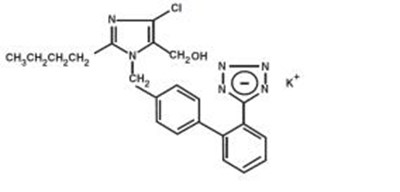
Figure 1 - losartan fig1

This text appears to be a comparison of the primary endpoint results for patients receiving Atenolol versus Losartan Potassium. An adjusted risk reduction of 13% was observed with Losartan Potassium compared to Atenolol at a significance level of p=0.021. The time period of the study spans over 66 months, with data collected at various time intervals from 6 to 66 months.*
Figure 3 - losartan fig3

This appears to be a table or data chart with demographic subgroups and their corresponding primary endpoint events. The table includes data on age, gender, race, history of VD, and other factors. Without additional context or information, it is unclear what the primary endpoint events refer to or what the significance of the data is.*
Figure-4 - losartan fig4

This seems to be a table displaying the results of a clinical trial comparing a medication called "Losttan Potassium" to a placebo. The data shows that out of the total number of patients, 60 patients had an event while taking Losttan Potassium, compared to 40 patients who had an event while taking the placebo. The risk reduction percentage is 16.1% and the results were statistically significant with a p-value of 0.022. It's unclear what type of event is being referenced.*
71205-194-90 - losartan fig7

This is a description of Losartan Potassium 100mg tablets with NDC 71205-194-90, distributed by Citron Pharma LLC. The tablets are green-colored, oval-shaped, and film-coated with 'E' marked on one side and '47' on the other. The product contains 90 tablets in a bottle, and each tablet contains Losartan Potassium USP 100mg. The medication should be stored at 20°-25°C (68°-77°F) out of the reach of children, and its expiration date is 00/00/00. The tablets were relabeled by Proficit Rx LP, Thousand Oaks, CA.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.