Product Images Tadalafil
View Photos of Packaging, Labels & Appearance
- Figure - tadalafil figure1
- Figure - tadalafil figure2
- Figure - tadalafil figure3
- Figure - tadalafil figure4
- Figure 5 - tadalafil figure5
- Figure 6 - tadalafil figure6
- Figure 7 - tadalafil figure7
- Figure 8 - tadalafil figure8
- 71205-268-30 - tadalafil label 20mg
- Chemical Structure - tadalafil structure
Product Label Images
The following 10 images provide visual information about the product associated with Tadalafil NDC 71205-268 by Proficient Rx Lp, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure - tadalafil figure1

The text seems to be a table showing the difference in blood pressure (measured in mmHg) between a group taking Tadalafil and a group taking Placebo. The table includes measurements for both standing and supine positions, as well as over a period of time (in hours). The text is not sufficient to provide further information.*
Figure - tadalafil figure2
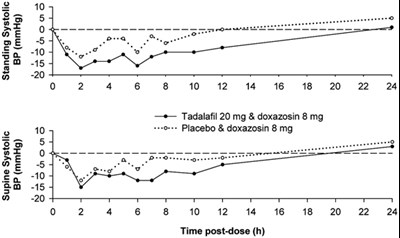
The text appears to be a combination of numerical values and some errors. The only useful information that can be extracted from the text is the title "Tadalafil 20mg & Doxazosin 8mg" along with a graph showing the effects of the medication over time. The rest of the text is either numerical data or errors, rendering it not usable.*
Figure - tadalafil figure4
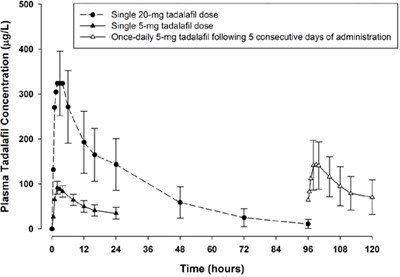
This is a graph displaying Plasma Tadalafil Concentration (in µg/L) over time (in hours) after administering different doses of Tadalafil. It shows the concentration for a single 20-mg dose, a single 5-mg dose, and a once-daily 5-mg dose following 5 consecutive days of administration. The concentration of Tadalafil decreases with time.*
Figure 5 - tadalafil figure5

The text provides information about the change from baseline in the IPSS total score of a medication called "tadalafils" compared to a placebo. The results show a statistically significant improvement in the IPSS total score for tadalafils compared to placebo (p<0.01) after 12 weeks of treatment.*
71205-268-30 - tadalafil label 20mg

This is a description of Tadalafil 20mg tablets with product ID QT026810. The tablets are yellow in color, almond-shaped, film-coated, and contain 20mg of Tadalafil USP per tablet. The tablets are embossed with "FJ4" on one side and plain on the other side. The tablets are packaged by Profcent Rx LP in Thousand Oaks, CA 81320. There are three different lots mentioned in this description, each with 10 tablets, labeled SNEMASTER, SN#MASTER, and SNAMASTER, with expiry date 00/00/00 and lot number 00000. The tablets are manufactured by Intas Pharmaceuticals Limited in Ahmedabad, India, and are RX Only. The NDC for the tablets is 71205-268-10 and the GTIN is 00371205268107. This medicine should be kept out of the reach of children and stored at 25°C (77°F).*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.




