Product Images Gabapentin
View Photos of Packaging, Labels & Appearance
- Formula - gabapentin fig1
- Figure 1 - gabapentin fig2
- Figure 2 - gabapentin fig3
- Figure 3 - gabapentin fig4
- Figure 4. Responder Rate in Patients Receiving Gabapentin Expressed as a Difference from Placebo by Dose and Study: Adjunctive Therapy Studies in Patients ≥12 Years of Age with Partial Seizures - gabapentin fig5
- 71205-373-15 - gabapentin fig9
- Chemical Structure - gabapentin str
Product Label Images
The following 7 images provide visual information about the product associated with Gabapentin NDC 71205-373 by Proficient Rx Lp, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure 1 - gabapentin fig2
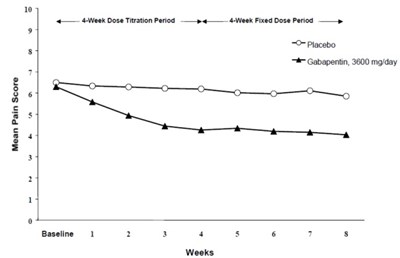
The data seems to be a graph displaying the mean pain score for different treatment periods using a placebo or the drug Gabapentin at a dosage of 3600 mg/day. The study had a baseline and lasted for 7 weeks. However, further information is not available due to the limited amount of text.*
Figure 2 - gabapentin fig3
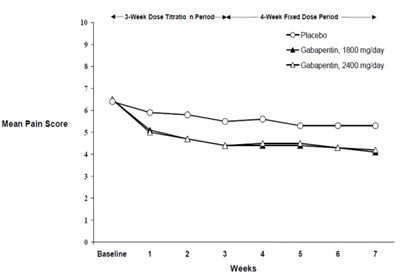
The text describes a table with different treatment periods and doses for managing pain. The table contains columns for mean pain score and baseline measurements. It includes three different treatment options; Placebo, Gabapentin 1800 mg/day, and Gabapentin 2400 mg/day. However, there is not enough information in the text to determine the type or cause of pain being studied.*
Figure 3 - gabapentin fig4
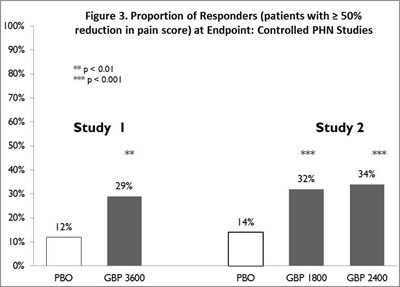
The text describes a graph named "Figure 3" that shows the proportion of responders in controlled PHN studies. The graph presents different percentages, including 100%, 70%, 50%, and 30%. Additionally, the graph shows the results of two studies labeled "Study 1" and "Study 2." Unfortunately, there is some corrupted text, and it is not possible to extract the full meaning from it.*
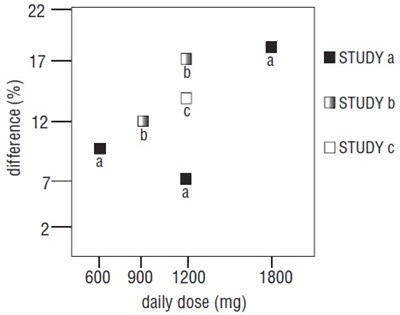
Figure 4. Responder Rate in Patients Receiving Gabapentin Expressed as a Difference from Placebo by Dose and Study: Adjunctive Therapy Studies in Patients ≥12 Years of Age with Partial Seizures - gabapentin fig5
71205-373-15 - gabapentin fig9

This is a description of Gabapentin 100mg capsules distributed by ProficientRx with the NDC 71205-373-15. The capsules are white and have "D" imprinted on the white cap and "02" on the white body. The capsules are filled with white to off-white crystalline powder. Each capsule contains 100mg of Gabapentin USP. The medication guide provided separately must be given to each patient. The medication should be stored at 20°-25°C (68°-77°F) and kept out of the reach of children. The product is packaged by Proficient Rx LP in Thousand Oaks, CA, and manufactured by Aurobindo Pharma USA, Inc. in India. The lot number of the product is 00000 and the expiration date is not available. The product ID is QG037315 and the GTIN is 00371205373153.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.
