Product Images Gabapentin
View Photos of Packaging, Labels & Appearance
- Figure 1 - ab1e0185 b802 4cf2 a5d4 b71f27f1dcb0 00
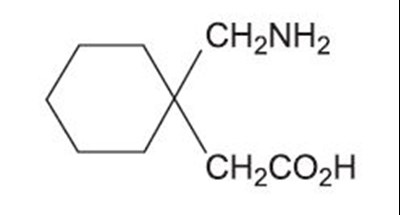
- Chem Structure - d45f499b 8612 479e 8dd2 4fe28b1f5f9c 01
- figure01 - d45f499b 8612 479e 8dd2 4fe28b1f5f9c 02
- figure02 - d45f499b 8612 479e 8dd2 4fe28b1f5f9c 03
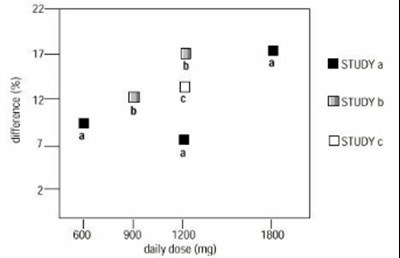
- figure03 - d45f499b 8612 479e 8dd2 4fe28b1f5f9c 04
- figure4 - d45f499b 8612 479e 8dd2 4fe28b1f5f9c 05
- 71205-457-90 - d45f499b 8612 479e 8dd2 4fe28b1f5f9c 06
Product Label Images
The following 7 images provide visual information about the product associated with Gabapentin NDC 71205-457 by Proficient Rx Lp, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure 1 - ab1e0185 b802 4cf2 a5d4 b71f27f1dcb0 00

This is a formula used to calculate the renal clearance of a patient based on their age, weight, and serum creatinine levels. The formula is adjusted for female patients by multiplying the result by 0.85.*
figure01 - d45f499b 8612 479e 8dd2 4fe28b1f5f9c 02

The text provides information on a measurement tool called "Moan Pain Score" and a 10 rating on it. Additionally, it mentions a 4-week dose titration period followed by a fixed dose period with Gabapentin at 3600 mg/day and a placebo. The text also includes a graph with weeks on the x-axis, but no further information is provided on it. Therefore, it can be concluded that the text provides limited information on a pain evaluation tool and a related medication trial.*
figure02 - d45f499b 8612 479e 8dd2 4fe28b1f5f9c 03

This is a chart outlining the pain scores for a 4-week fixed dose period. It includes a placebo group, as well as groups taking two different doses of Gabapentin (1800mg/day and 2400mg/day). However, without additional information, it is unclear what type of pain is being measured and how the mean scores differ between the groups over the 7-day period.*
figure03 - d45f499b 8612 479e 8dd2 4fe28b1f5f9c 04

The figure shows the proportion of patients who experienced a reduction in pain score greater than 50% at the end of two controlled studies on post-herpetic neuralgia (PHN). The results of Study 1 are not provided. The percentage of responders in Study 2 was 29% for GBP 800 and 32% for GBP 2400. The asterisk (*) denotes statistical significance, but the exact p-value is not readable. The remaining text may refer to dosages or costs associated with GBP (presumably gabapentin).*
71205-457-90 - d45f499b 8612 479e 8dd2 4fe28b1f5f9c 06

This is a description of a medication called Gabapentin 600mg. It is an oval-shaped tablet that is white or off-white with a film coating. Each tablet is scored on both sides and has "NT* and *150" on one side. It is dispensed in a bottle of 90 tablets with a separate medication guide for each patient. The medication is manufactured by Mis STRIDES SHASUN LIMITED, UNIT-Il R S No 32,33 & 34, PIMS Road, Periyakalapet, Puducherry-605 014, India and packaged by Proficent Rx LP, Thousand Oaks, CA 91320. The medication should be kept between 20°-25°C (68°-77°F) and stored out of the reach of children. The expiration date and lot number are also included in the description.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.