Product Images Selegiline Hydrochloride
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 3 images provide visual information about the product associated with Selegiline Hydrochloride NDC 71205-888 by Proficient Rx Lp, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
71205-888-30 - 500ct

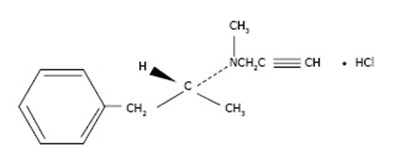
This is a prescription drug labeled Selegiline HCI 5mg packaged by Proficient Rx LP. It comes in a bottle of 30 hard gelatin capsules with an opaque white body and blue opaque cap with the imprint "P 659" in black ink on both cap and body. Each capsule contains Selegiline Hydrochloride, USP 5 mg. It is distributed by Rising Pharma Holdings, Inc. East Brunswick, NJ 08816 and made in the USA with an ID of Q5088830. It is important to store this medicine at 20°-25°C (68°-77°F) and out of the reach of children.*
adverse reactions table - table adverse events v2

This is a report of the incidence of adverse experiences encountered during placebo-controlled clinical trials of selegiline hydrochloride conducted on a sample of 50 patients. The adverse events reported include nausea, dizziness, lightheadedness, fainting, abdominal pain, confusion, hallucinations, dry mouth, dyskinesias, headache, vivid dreams, anxiety/tension, anemia, diarrhea, hair loss, insomnia, leg pain, low back pain, malaise, palpitations, urinary retention, lethargy, and weight loss.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.