Product Images Miglitol
View Photos of Packaging, Labels & Appearance
- 4000882 Miglitol 25mg (WP)(v.1) 201028 for elisting
- 4000883 Miglitol 50mg (WP)(v.1) 201028 for elisting
- 4000884 Miglitol 100mg (WP)(v.1) 201028 for elisting
- 1c5a6b7-900b-4eff-ac99-1df08fa33854 - 41c5a6b7 900b 4eff ac99 1df08fa33854
- abcdea2-1f08-4f44-bac7-e6a43bc5a32f - eabcdea2 1f08 4f44 bac7 e6a43bc5a32f
Product Label Images
The following 5 images provide visual information about the product associated with Miglitol NDC 71205-935 by Proficient Rx Lp, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
4000882 Miglitol 25mg (WP)(v.1) 201028 for elisting
(v.1)_201028 for elisting.jpg.jpg.jpg)
Miglitol 25mg is a medication available in the form of film-coated tablets containing 25 mg of miglitol. The tablets are white to off-white, circular, biconvex film-coated, debossed with the logo-mark "OP* on one side and the product code "25” on the other side. This medication is manufactured by Mir By Onent Pharma Co, Ltd, located in Yuniin, Taiwan, and is packaged by Proficient Rx LP, located in Thousand Oaks, CA. The product comes in a bottle that contains 30 tablets and has a Product ID: QM093530, NDC: 71205-935-30, and GTIN: 0371205935306. The medication should be stored at 25°C (77°F) and kept out of the reach of children. The expiry date and the lot number of the product are not available. This medication requires a prescription.*
4000883 Miglitol 50mg (WP)(v.1) 201028 for elisting
(v.1)_201028 for elisting.jpg.jpg.jpg)
ProficientRx is a manufacturer of Miglitol 50mg in pack of 30 tablets. Each tablet is film-coated with 50mg of Miglitol and has the logo-mark "OP" on one side and the product code "*26" on the other side. The product is manufactured by Orient Pharma Co, Ltd in Yunin, Taiwan and is to be stored at 25°C (77°F). The medication is available only through a prescription and the product ID is QM093630. The packaging is done by Proficient Rx LP in Thousand Oaks, CA 91320 with NDC 71205-936-30 and GTIN 0371205936303. The SN# MASTER, Lot#: 00000 and Exp. 00/00/00 details are also provided.*
4000884 Miglitol 100mg (WP)(v.1) 201028 for elisting
(v.1)_201028 for elisting.jpg.jpg.jpg)
Miglitol 100mg tablets are a prescription medication that comes in a white to off-white, circular, biconvex film-coated tablet form. The tablets are debossed with the logo mark "OP" on one side and product code *27* on the other side. Each film-coated tablet contains 100 mg of miglitol. The medication is made by Orient Pharma Co., Ltd. and packaged by Proficient Rx LP in Thousand Oaks. The medication should be stored at 25°C (77°F), and the expiry date is 00/00/00 with lot #00000. The GTIN is 00371205937300. It is essential to keep the drug out of the reach of children.*
1c5a6b7-900b-4eff-ac99-1df08fa33854 - 41c5a6b7 900b 4eff ac99 1df08fa33854
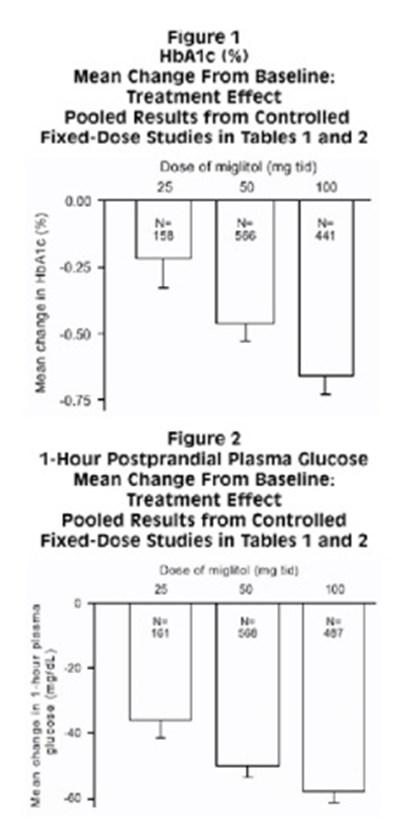
The text describes figures 1 and 2 featuring mean change from baseline and treatment effect. It presents the pooled results from controlled fixed-dose studies in tables 1 and 2 concerning 5 HBATE. Additionally, figure 2 shows the treatment effect on postprandial plasma glucose after 1 hour.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.
