Product Images Donepezil Hydrochloride
View Photos of Packaging, Labels & Appearance
- 01 - 30406488 figure 01
- figure2 - 30406488 figure 02
- figure-03 - 30406488 figure 03
- figure-04 - 30406488 figure 04
- figure-05 - 30406488 figure 05
- figure-06 - 30406488 figure 06
- figure-07 - 30406488 figure 07
- 08 - 30406488 figure 08
- 09 - 30406488 figure 09
- 10 - 30406488 figure 10
- 11 - 30406488 figure 11
- 12 - 30406488 figure 12
- 13 - 30406488 figure 13
- 14 - 30406488 figure 14
- 15 - 30406488 figure 15
- 71205-948-30 - 30406488 figure 16
Product Label Images
The following 16 images provide visual information about the product associated with Donepezil Hydrochloride NDC 71205-948 by Proficient Rx Lp, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
figure2 - 30406488 figure 02
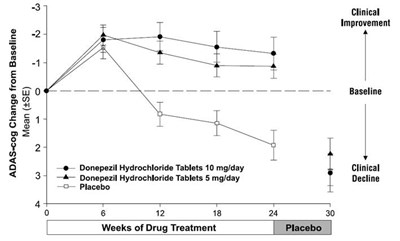
This is a graph showing the ADAS-cog change from baseline over 24 weeks of drug treatment with Donepezil Hydrochloride Tablets 10mg/day, Donepezil Hydrochloride Tablets 5mg/day, and Placebo. There are two lines depicting clinical improvement and clinical decline observed in patients.*
figure-03 - 30406488 figure 03
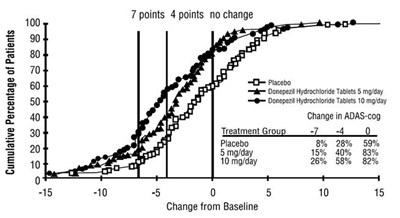
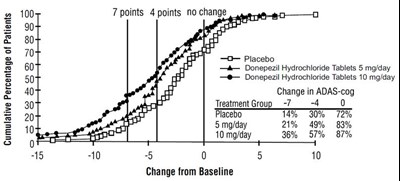
The text describes a chart showing the cumulative percentage of patients in different treatment groups (placebo, Donepezil Hydrochloride Tablets 5mg/day, Donepezil Hydrochloride Tablets 10mg/day) with various levels of change in ADAS-cog. The chart also shows the corresponding change from baseline.*
figure-04 - 30406488 figure 04

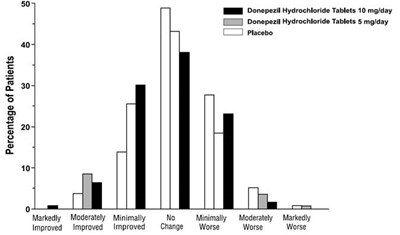
This text provides data on the percentage of patients who took either Donepezil Hydrochloride Tablets (10 mg/day or 5 mg/day) or Placebo. The results are presented on a graph which shows the percentage of patients who experienced Markedly, Moderately, Minimally or No Improvement in their CIBIC-plus Rating after taking the medication.*
figure-05 - 30406488 figure 05

This is a table showing the ADAS-cog change from baseline (in mean+SE) for three treatments- Donepezil Hydrochloride Tablets 10mg/day, Donepezil Hydrochloride Tablets 5mg/day and Placebo across 5 weeks of drug treatment. The table also indicates clinical improvement.*
08 - 30406488 figure 08

This appears to be a table display of some clinical trial data. The label "SIB Change from Baseline" suggests that the study evaluated the change in a score called SIB over time. The table shows the mean score (+SE) for two different treatments: "Donepezil Hydrochloride Tablets 10 mg/day" and "Placebo". "Clinical Improvement" and "Baseline" may refer to different cohorts or time points. The study appears to have lasted for 3 months.*
10 - 30406488 figure 10

This appears to be a graph showing the change in ADCS-ADL scores over three months of drug treatment with either placebo or Donepezil Hydrochloride Tablets 10 mg/day. The graph shows clinical improvement for Donepezil Hydrochloride Tablets compared to a baseline and clinical decline with placebo.*
11 - 30406488 figure 11
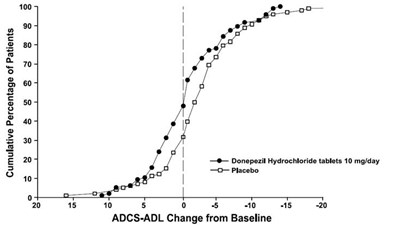
The text describes a chart or graph that measures the change in ADCS-ADL (Alzheimer's Disease Cooperative Study-Activities of Daily Living) from Baseline in patients taking Donepezil Hydrochloride tablets at a dose of 10 mg/day and in patients taking placebo. The chart shows values ranging from -20 to 20 along the x-axis and the treatments along the y-axis. The symbol "®" appears before the number 100, followed by two spaces and the gibberish text "oo". It is not clear what this symbol and text represent.*
13 - 30406488 figure 13

The graph shows the cumulative percentage of patients taking different doses of Donepezil Hydrochloride Tablets and their corresponding change from baseline on the SIB scale. The X-axis represents the change in the SIB score, and the Y-axis represents the percentage of patients. There are two doses shown: 10mg/day and 23mg/day. The data shows that a larger percentage of patients taking 23mg/day had positive changes in SIB score compared to those taking 10mg/day.*
14 - 30406488 figure 14

This appears to be a table or chart with different medications and their effects on patients, including improvement and changes in rating. The medications listed are Donepezil Hydrochloride Tablets, with one group receiving 23mg per day and another group receiving 10mg per day. The chart includes percentages of patients in different categories of improvement and change, as well as a CIBIC-plus rating. Without more context, it is not possible to say what specific condition or disorder these medications are intended for.*
71205-948-30 - 30406488 figure 16

This is a description of Donepezil HCI 23mg tablets. The tablets are white and film-coated with round shape. The tablets are printed with “T004" in black ink on one side and plain on the other side. Each tablet contains Donepezil Hydrochloride, 23 mg. The product is manufactured by TWI Pharmaceuticals, located in Taoyuan City, 32063, Taiwan. The tablets come in a bottle of 30 and are RX-only. The medication should be stored between 20°-25°C (68°-77°F) and kept out of the reach of children. The product was relabeled by Proficient Rx LP, located in Thousand Oaks, CA 91320.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.