Product Images Donepezil Hydrochloride
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 20 images provide visual information about the product associated with Donepezil Hydrochloride NDC 71209-019 by Cadila Pharmaceuticals Limited, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
donepezil-spl-cont-label-10mg-30tab - donepezil spl cont label 10mg 30tab

This is a description of a medication that contains Dorl hydrochloride and Donepezil Hydrochloride in tablet form. The tablets are coated and must be stored in a controlled room temperature between 59°F to 86°F (15°C to 30°C). They should be dispensed in tight containers and the packaging includes dosage and usage instructions. The manufacturer is CADILA PHARMACEUTICALS LTD in India.*
donepezil-spl-cont-label-10mg-90tab - donepezil spl cont label 10mg 90tab

Each film-coated tablet contains 10 mg of Donepezil Hydrochloride USP. This medication is prescribed to treat dementia related to Alzheimer's disease. The tablets should be stored at temperatures between 15°C and 30°C. The medication should be dispensed in tight containers that conform to the USP standards. It is manufactured by Cadila Pharmaceuticals Ltd. in India.*
donepezil-spl-cont-label-5mg-1000tab - donepezil spl cont label 5mg 1000tab

This is a medication description for Donepezil Hydrochloride Tablets USP, manufactured by Cadila Pharmaceuticals Ltd. The tablets are coated and each tablet contains 5 mg of Donepezil Hydrochloride USP. The dosage and usage information must be obtained from the prescribing information. The tablets should be stored in tight containers at room temperature between 59°F to 86°F. The product is only available on prescription and comes in a pack of 1000 tablets. The National Drug Code (NDC) for this product is 71209-019-11.*
donepezil-spl-cont-label-5mg-30tab - donepezil spl cont label 5mg 30tab

Each tablet of Donepezil hydrochloride USP 5mg is film-coated and has the NDC number 71209-019-01. The recommended dosage and instructions for use can be found in the accompanying prescribing information. The tablets should be stored at a controlled room temperature between 59° to 86°F (15° to 30°C). They should only be dispensed in USP containers. The manufacturer is CADILA PHARMACEUTICALS LTD. located in Bhid 1389, Dhlka, Dist, Ahmedabid, Gujarat, DA.*
donepezil-spl-cont-label-5mg-90tab - donepezil spl cont label 5mg 90tab

Each film-coated tablet of Donepezi Hydrochloride USP 5mg contains NG71203-019-04. It is a prescription medication used for treatment and should be used as per the prescribing information. Store in a cool and dry place between 15 to 30 degree Celsius, in a tight container, and out of reach of children. Manufactured by Cadila Pharmaceuticals Ltd. in India.*
fig3 - donepezil spl fig 03
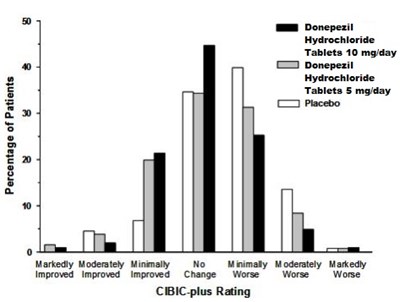
This is a table that displays information about Donepezil Hydrochloride Tablets dosage of 10 mg/day and 5 mg/day. There is also an option for Placebo. The table also includes a rating scale from Markedly Improved to Worse using CIBIC plus Rating.*
fig6 - donepezil spl fig 06

This appears to be a chart or graph showing the percentage of patients who experienced different levels of improvement after taking either Donepezil Hydrochloride tablets at 10mg/day or 5mg/day. The chart shows different levels of improvement, ranging from markedly improved to markedly worse, as well as the percentage of patients who experienced each level of improvement. There is also a CIBIC-plus rating included.*
fig8 - donepezil spl fig 08

This is a table representing the SIB (Severe Impairment Battery) change from baseline for three different treatments: Donepezil Hydrochloride, Placebo, and Tablets 10 mg/day. The table shows numerical values for each treatment option, but there is no additional information available to provide context or further explanation.*
fig13 - donepezil spl fig 13

This is a data table showing the percentage of patients who experienced improvement or worsened based on their CIBIC-plus rating when taking Donepezil Hydrochloride tablets in varying dosages. Information on the dosages administered is also provided.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.










