Product Images Heparin Sodium
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 15 images provide visual information about the product associated with Heparin Sodium NDC 71288-403 by Meitheal Pharmaceuticals Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
hep0d 0005 02

This is a description for Heparin Megheal Sodium Injection, which is meant for intravenous or subcutaneous use. It is a single-dose vial that is not intended for lock flush. It contains USP units per mL of heparin, derived from porcine intestinal mucosa, and is manufactured for Meitheal Pharmaceuticals in Chicago, IL. The product is only available by prescription.*
hep0d 0005 03

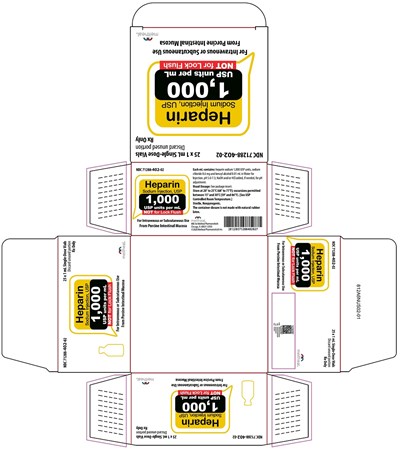
This text describes a medication called "Sodium Injection USP" manufactured by Meitheal Pharmaceuticals. The medication comes in a multi-dose vial containing 10 ml, with a strength of 1,000 USP units per mL. It is intended for intravenous or subcutaneous use only and should not be used for lock flush.*
hep0d 0005 04
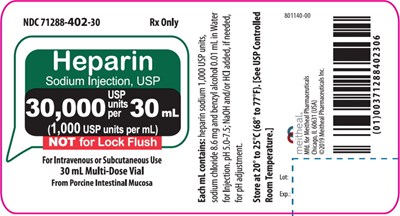
This appears to be a mix of various texts, including a financial amount in Euros, a code for a product, and information on a medical product used for intravenous or subcutaneous injection. There is also some gibberish characters and symbols on the page which could not be interpreted correctly.*
hep0d 0005 05

This is a description of a medication called Heparin Sodium Injection, USP, with a concentration of 5,000 USP units per mL. The active ingredient (heparin) is obtained from porcine intestinal mucosa. The medication is manufactured by Meitheal Pharmaceuticals in Chicago, IL, and is available in a single-dose vial for intravenous or subcutaneous use only. The medication is not meant for lock flush, as stated on the label. The NDC number for this medication is 71288-403-01 and it is marked for prescription only. The year of manufacture for this medication is 2019.*
hep0d 0005 06

This is a Sodium Injection USP available in a 10 mL Multi-Dose Vial. It contains 5,000 USP units of heparin sodium per mL along with sodium chloride and benzyl alcohol in water. It is intended for intravenous or subcutaneous use and should not be used for lock flush. It may require pH adjustment using NaOH or HCl. It should be stored at 20° to 25°C (68° to 77°F) and was manufactured by Meitheal Pharmaceuticals in Chicago, IL in 2019.*
hep0d 0005 07

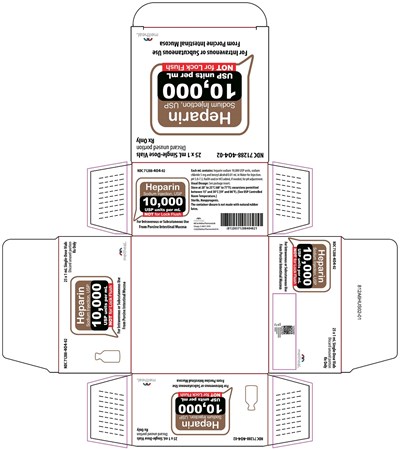
This is a product label for the medication "Sodium Injection, USP" which is administered intravenously or subcutaneously. It contains 10,000 USP units per milliliter and is packaged in single-dose vials. The medication is manufactured by Meitheal Pharmaceuticals in Chicago, USA and is not for lock flush.*
hep0d 0005 08

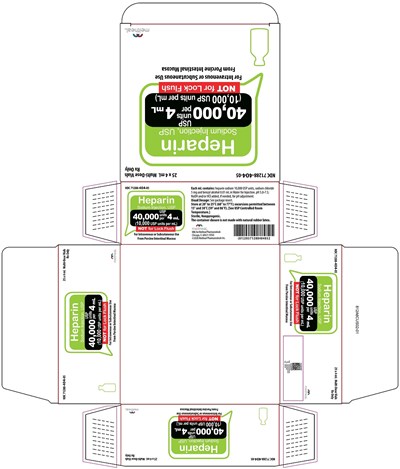
This appears to be a label of a medication with the NDC number 71288-404-04. The medication can only be obtained with a prescription. Other information like manufacturer name and address, dosage, and packing can be seen but the result is not fully reliable.*
hep0d 0005 13 v2
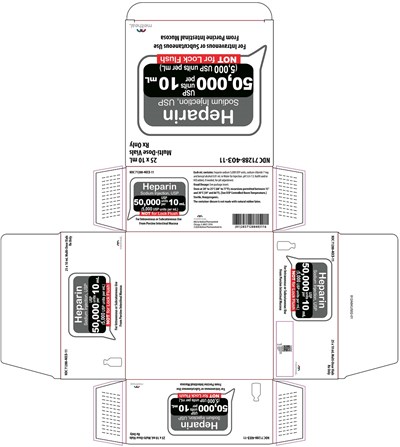
This is a medical label for Heparin Sodium Injection, USP. The label indicates that the product is not suitable for Lock Flush and provides information on the dosage and other instructions for use. The text also includes a product code and lot number.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.