Product Images Fosinopril Sodium
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 4 images provide visual information about the product associated with Fosinopril Sodium NDC 71335-0187 by Bryant Ranch Prepack, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
image - 9d869f7c 9b5a 47aa 9a82 35a0f86112b2 02
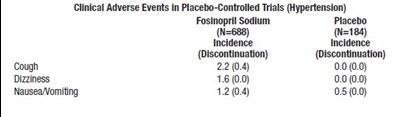
This text shows the incidence of clinical adverse events in placebo-controlled trials for hypertension with Fosinopril Sodium and Placebo. The table includes the number of participants for each group (688 for Fosinopril Sodium and 184 for placebo), and the incidence of adverse events such as cough, dizziness, nausea and vomiting, as well as the number of participants who discontinued the trial due to these adverse events.*
Label Image - lbl713350187

This is a description of a medication containing Fosinopril and Sodium used for treating cardiovascular diseases. The medication is in form of a round, white tablet with imprint code 1G;201, and it comes in a package of 30 tablets. The medication should be stored at room temperature, between 20°C-25°C (68°F-77°F), and kept out of reach of children. The medication is manufactured by InvaGen Pharmaceuticals, Inc., and its NDC code is 7133501871 06994301523487.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.

