Product Images Donepezil Hydrochloride
View Photos of Packaging, Labels & Appearance
- str - de82e6d7 f231 449a a2aa 91e26eb50774 01
- fig.1 - de82e6d7 f231 449a a2aa 91e26eb50774 02
- Figure 2 - de82e6d7 f231 449a a2aa 91e26eb50774 03
- Figure 3 - de82e6d7 f231 449a a2aa 91e26eb50774 04
- 5 - de82e6d7 f231 449a a2aa 91e26eb50774 05
- figure 5 - de82e6d7 f231 449a a2aa 91e26eb50774 06
- figure 6 - de82e6d7 f231 449a a2aa 91e26eb50774 07
- Figure 7 - de82e6d7 f231 449a a2aa 91e26eb50774 08
- Figure 8 - de82e6d7 f231 449a a2aa 91e26eb50774 09
- Figure 9 - de82e6d7 f231 449a a2aa 91e26eb50774 10
- Figure 10 - de82e6d7 f231 449a a2aa 91e26eb50774 11
- logo - de82e6d7 f231 449a a2aa 91e26eb50774 12
- logo - de82e6d7 f231 449a a2aa 91e26eb50774 13
- Label Image - lbl713350582
Product Label Images
The following 14 images provide visual information about the product associated with Donepezil Hydrochloride NDC 71335-0582 by Bryant Ranch Prepack, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
fig.1 - de82e6d7 f231 449a a2aa 91e26eb50774 02

This is a chart showing the change in ADAS-cog score from baseline, with mean values represented by symbols for Donepezil Hydrochloride Tablets 10mg/day, Donepezil Hydrochloride Tablets 5mg/day, and Placebo. The chart also shows clinical improvement and clinical decline from baseline, with weeks of drug treatment shown along the X-axis.*
Figure 2 - de82e6d7 f231 449a a2aa 91e26eb50774 03

This represents a graph displaying the cumulative percentage of patients with a change in ADAS-cog score while being treated with Donepezil Hydrochloride Tablets 5mg/day, Donepezil Hydrochloride Tablets 10mg/day, or a placebo. The x-axis represents the change from baseline score, while the y-axis shows the percentage of patients. The treatment group had a significantly higher percentage of patients experiencing cognitive improvement than the placebo group.*
5 - de82e6d7 f231 449a a2aa 91e26eb50774 05

This is a graph that shows the ADAS-cog (Alzheimer's Disease Assessment Scale - cognitive subscale) Change from Baseline over 12 weeks of drug treatment with different doses of Donepezil Hydrochloride (5mg/day and 10mg/day) and placebo. The y-axis shows the mean change score and the x-axis shows the weeks of treatment. The graph also shows clinical improvement and clinical decline, and it appears to have been a study testing the effectiveness of the different doses of Donepezil Hydrochloride in treating Alzheimer's disease.*
figure 5 - de82e6d7 f231 449a a2aa 91e26eb50774 06

This is a chart showing the cumulative percentage of patients in different treatment groups with Alzheimer's disease and their change in ADAS-COG (a cognitive assessment) score from baseline. The treatment groups include 5mg/day and 10mg/day of Donepezil Hydrochloride tablets as well as a placebo group. The data shows that those in the 10mg/day group had the highest percentage of improved ADAS-COG scores, with 87% showing improvement after 24 weeks of treatment.*
figure 6 - de82e6d7 f231 449a a2aa 91e26eb50774 07
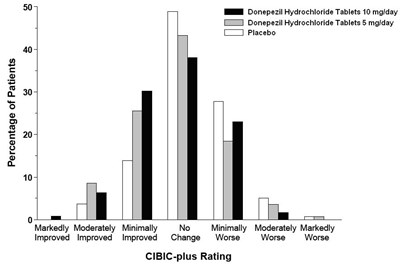
This is a graph showing the percentage of patients who have experienced improvement or worsening changes in their CIBIC-plus rating while taking BN donepezil hydrochloride tablets, donepezil hydrochloride tablets or a placebo. The dosages mentioned are 10 mg/day and 5mg/day. The graph also shows four levels of improvement and worsening change in the patients' condition. There is not enough information to determine what the CIBIC-plus rating is or what it is used for.*
Figure 8 - de82e6d7 f231 449a a2aa 91e26eb50774 09
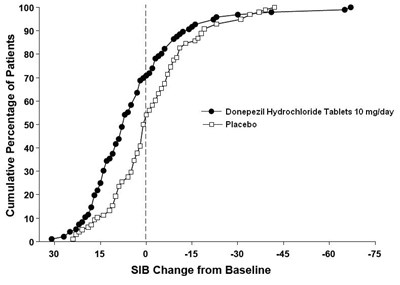
The text appears to be a graph showing the cumulative percentage of patients and their SIB (Severe Impairment Battery) change from baseline. The graph compares patients taking Donepezil Hydrochloride Tablets (10 mg/day) to those taking a placebo. There is a cumulative percentage of 100 at the top of the graph with SIB change from baseline on the Y-axis.*
Figure 9 - de82e6d7 f231 449a a2aa 91e26eb50774 10

This is a figure showing the Time Course of the Change from Baseline in ADCS-ADL-Severe Score for Patients Completing 6 Months of Treatment. It compares the clinical improvement and decline in patients who were administered with Donepezil Hydrochloride Tablets 10mg/day versus Placebo over a period of 6 months. The X-axis represents the months of drug treatment while the Y-axis represents the change from baseline in ADCS-ADL-Severe Score.*
Figure 10 - de82e6d7 f231 449a a2aa 91e26eb50774 11

This text is a graph that shows the cumulative percentage of patients for Donepezil Hydrochloride Tablets 10 mg/day and placebo. The x-axis shows the change from baseline in ADCS-ADL, while the y-axis represents the cumulative percentage of patients. There are no further details provided in the text.*
Label Image - lbl713350582

This is a medicine package for Donepezil HCI yellow 10mg tablets made by Torrent Pharmaceuticals Limited. The tablets are yellow and round in shape, and are stored at room temperature between 20-25°C (68°-77°F). The package includes 30 tablets and the product is compared to Aricept 10mg tablets. The expiration date is not provided, but the product has an NDC number of 7133505821 and a UPC code of 05219301523487.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.




