Product Images Topiramate
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 4 images provide visual information about the product associated with Topiramate NDC 71335-1143 by Bryant Ranch Prepack, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
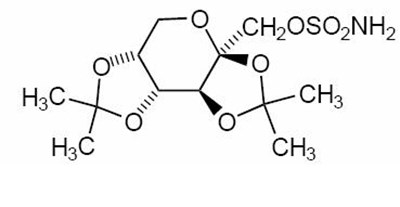
Structured product formula for Topiramate - 39703b05 f774 4aed 9792 000a61531553 02
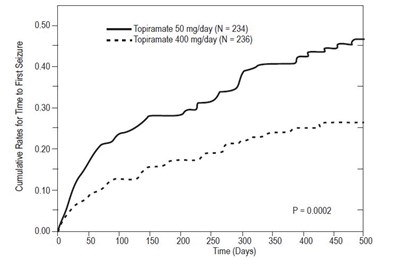
This is a graph displaying cumulative rates for time to first seizure in patients administered with Topiramate 50 mg/day (N = 234) and Topiramate 400 mg/day (N = 236). It shows the probability of experiencing a seizure during different time intervals and indicates that the group taking Topiramate 400 mg/day has a significantly lower rate of seizures.*
Label - lbl713351143

The packaging information indicates that the medicine, Topiramate, in the form of white round tablets, has been prepacked by Bryant Ranck Prepack Bunbauk in the quantity of 60 tablets per package. Each 100mg tablet is meant to be stored at room temperature, specifically between 20°C-25°C (68°F-77°F), and should be kept out of the reach of children. The National Drug Code (NDC) for this pharmaceutical item is 7133511431, and the expiration date is not available.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.