Product Images Gabapentin
View Photos of Packaging, Labels & Appearance
- image - d3fd11fe 4d3f 4002 8f9e 42cc12b4252d 01
- image - d3fd11fe 4d3f 4002 8f9e 42cc12b4252d 03
- image - d3fd11fe 4d3f 4002 8f9e 42cc12b4252d 04
- image - d3fd11fe 4d3f 4002 8f9e 42cc12b4252d 05
- image - d3fd11fe 4d3f 4002 8f9e 42cc12b4252d 06
- image - d3fd11fe 4d3f 4002 8f9e 42cc12b4252d 07
- image - d3fd11fe 4d3f 4002 8f9e 42cc12b4252d 08
- image - d3fd11fe 4d3f 4002 8f9e 42cc12b4252d 09
- image - d3fd11fe 4d3f 4002 8f9e 42cc12b4252d 10
- image - d3fd11fe 4d3f 4002 8f9e 42cc12b4252d 11
- image - d3fd11fe 4d3f 4002 8f9e 42cc12b4252d 12
- Label Image - lbl713351454
Product Label Images
The following 12 images provide visual information about the product associated with Gabapentin NDC 71335-1454 by Bryant Ranch Prepack, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
image - d3fd11fe 4d3f 4002 8f9e 42cc12b4252d 01

This is a dosage table for gabapentin based on renal function. The table shows the total daily doses for different creatinine clearance levels. The dosage regimen varies from 300TID to 1,200TID for patients with creatinine clearance >60 mL/min, while for patients with creatinine clearance <15 mL/min, the daily dose should be reduced proportionally. Patients on hemodialysis should receive maintenance doses based on creatinine clearance estimates and a supplemental post-hemodialysis dose. The Cockcroft and Gault equation can be used to estimate creatinine clearance.*
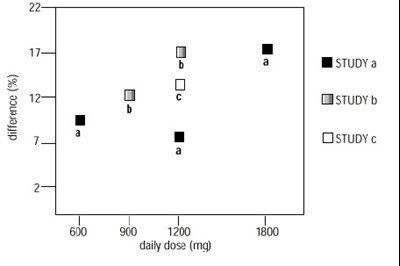
image - d3fd11fe 4d3f 4002 8f9e 42cc12b4252d 03
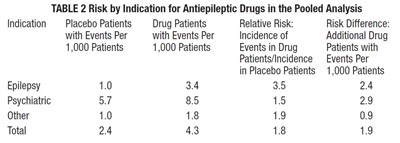
This text provides a table (table 2) comparing the risks of antiepileptic drugs for different indications (epilepsy, psychiatric, and other) versus placebo patients. The table provides data on the number of patients with events per 1,000 patients for both placebo and drug patients, as well as the relative risk and risk difference.*
image - d3fd11fe 4d3f 4002 8f9e 42cc12b4252d 04

This is a report on Adverse Reactions in Pooled Placebo-Controlled Trials in Postherpetic Neuralgia. The table shows the percentage of adverse reactions experienced by patients under Gabapentin (N=336) compared to those under Placebo (N=227). The adverse reactions are categorized according to Body as a Whole, Digestive System, Metabolic and Nutritional Disorders, Nervous System, Respiratory System, and Special Senses. The adverse reactions reported include Asthenia, Infection, Accidental injury, Diarrhea, Dry mouth, Constipation, Nausea, Vomiting, Peripheral edema, Weight gain, Hyperglycemia, Dizziness, Somnolence, Ataxia, Abnormal thinking, Abnormal gait, Incoordination, Pharyngitis, Amblyopia, Conjunctivitis, Diplopia and Otitis media. There is a note on blurred vision reported in the asterisk.*
image - d3fd11fe 4d3f 4002 8f9e 42cc12b4252d 05

The text provides a table of adverse reactions in epilepsy patients greater than 12 years of age, detailing the percentage of patients who experienced side effects while taking Gabapentin and a placebo. The adverse reactions are listed under different body systems such as body as a whole, cardiovascular, digestive system, nervous system, respiratory system, skin and appendages, urogenital system, and special senses. Some of the adverse reactions mentioned include fatigue, increased weight, dyspepsia, somnolence, dizziness, pharyngitis, coughing, diplopia, amblyopia, and more.*
image - d3fd11fe 4d3f 4002 8f9e 42cc12b4252d 06

This is a table showing the adverse reactions reported in a placebo-controlled add-on trial involving pediatric epilepsy patients aged 3 to 12 years who were given Gabapentin. The table compares adverse reaction incidence for Gabapentin compared to Placebo. The most commonly reported adverse reactions during the trial were fever, somnolence, hostility, nausea and/or vomiting, along with emotional lability.*
image - d3fd11fe 4d3f 4002 8f9e 42cc12b4252d 08

This is a table showing the duration of two controlled PHN studies, the dosages given to patients, and the number of patients receiving gabapentin and placebo. The target dose for Study 1 was 3,600 mg/day, while for Study 2, it was 1,800 and 2,400 mg/day. The total number of patients receiving gabapentin was 336, while those receiving placebo were 227. Dosages were given in three divided doses per day (TID).*
Label Image - lbl713351454

This appears to be a packaging label for a prescription drug called "Neurontin 300mg" made by InvaGen Pharmaceuticals. It includes information such as the dosage form (capsule), quantity per package (#90), and expiration date (MM/YY). It also provides the National Drug Code (NDC) and storage instructions. The label includes a warning to keep all drugs out of reach of children.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.