Product Images Oxycodone And Acetaminophen
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 4 images provide visual information about the product associated with Oxycodone And Acetaminophen NDC 71335-1472 by Bryant Ranch Prepack, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Label - lbl713351472

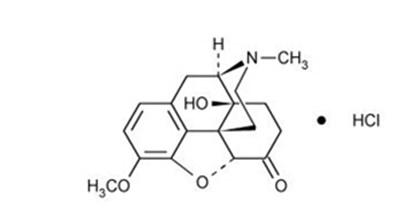
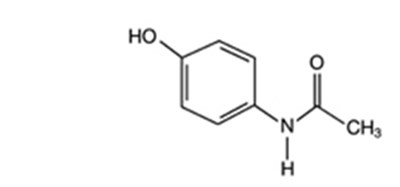
This is a description of Oxycodone Hydrochloride and Acetaminophen tablets which contain 7.5 mg of oxycodone and 325 mg of acetaminophen. The medication comes in tablet form and is intended for prescription use only, carrying the risk of overdose and addiction. The tablets may cause drowsiness and should be stored under specific temperature conditions. A National Drug Code (NDC) is provided along with the number of tablets in each package.*
Label - lbl7133514722

This appears to be a label or packaging information for a medication called "Percocet" manufactured by Purdue Pharma L.P. The medication is in the form of tablets containing 7.5mg oxycodone and 325mg acetaminophen. The package was prepared by Bryant Ranck as a prepack, and the label also includes information on storage temperature and a warning of possible drowsiness as a side effect. The label has an NDC code of 7133514721 and an expiration date of MM/YY.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.