Product Images Venlafaxine Hydrochloride
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 7 images provide visual information about the product associated with Venlafaxine Hydrochloride NDC 71335-1868 by Bryant Ranch Prepack, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
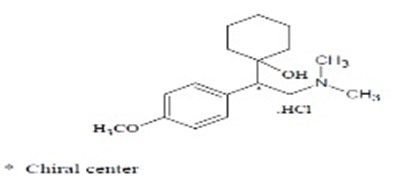
figr1

This text appears to be a list of interacting drugs and their effects on analytes like venlafaxine, along with recommendations for PK Fold Change and 90% CI. The list also includes advice on avoiding concomitant use or using with caution in certain patients.*
figr2

This is a drug interaction table showing the effects of various drugs on each other when taken concomitantly. The table lists drugs such as ethanol, diazepam, alprazolam, indinavir, metoprolol, and lithium, among others, and the recommended dose adjustments, if any. The table lists fold changes in PK (pharmacokinetics) and 90% CI (confidence interval). Additionally, the table mentions drugs that should be used with caution, and some should be avoided if possible.*
figr3

This is a chart or table indicating recommendations for dosage adjustments for the drug venlafaxine based on specific patient populations, including those with hepatic impairment (cirrhosis, mild or moderate impairment), renal impairment (hemodialysis), CYP2D6 polymorphism, age (>60 years), and gender. The chart suggests reductions in dose for most of the patient populations mentioned, with some recommendations for individualization of dosing. The chart also includes information on changes in PK (pharmacokinetics) and fold change relative to reference values.*
Label - lbl713351868

This is a medication description for Venlafaxine HCL 75mg extended-release capsules manufactured by Annora Pharma Pvt. Ltd. They are pink and white in color and may cause drowsiness. The capsules should be stored at room temperature and kept away from children. The package contains 90 capsules and has an expiration date of MM/YY. The NDC number for this medication is 7133518681 03325901523487. The rest of the text appears to be a label from a supplier or distributor and is not relevant to the medication description.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.