Product Images Aripiprazole
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 11 images provide visual information about the product associated with Aripiprazole NDC 71335-1892 by Bryant Ranch Prepack, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure1 - Figure1
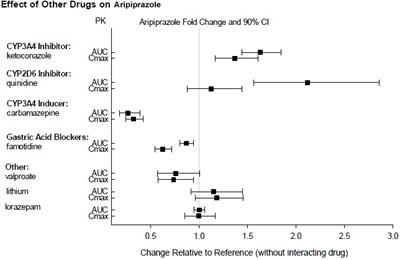
This is a table showing the effect of various drugs on aripiprazole, with fold change and 90% C1 values. The drugs include inhibitors and inducers of certain cytochrome P450 enzymes, gastric acid blockers, and other drugs like valproate and lithium. The table also provides a relative change in aripiprazole levels compared to a reference value without the interacting drug.*
Figure2 - Figure2
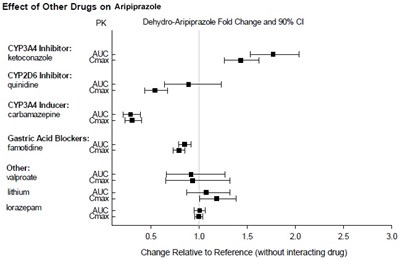
This text reports the effect of several drugs on Aripiprazole, an antipsychotic medication. The drugs are classified based on their effect on certain liver enzymes, such as CYP3A4 inhibitor ketoconazole or CYP2D6 inhibitor quinidine. The text also mentions the effect of gastric acid blockers like famotidine and other drugs like valproate and lorazepam. The effects are measured by the Fold Change and 90% CI of Dehydro-Aripiprazole. There is a graph showing a change relative to reference without any interacting drug.*
Figure3 - Figure3

The text contains information on the effect of Aripiprazole on other drugs but it is not clear due to some misspelled words, no context and lack of relevant information. Therefore, the description cannot be generated accurately.*
Figure4 - Figure4
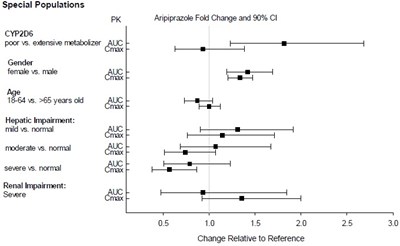
This text seems to be a list of parameters related to pharmacokinetics (PK) and drug metabolism in special populations. The parameters include genetic differences in CYP2D6 metabolism ("poor" vs. "extensive" metabolizers), gender, age, and hepatic and renal impairment. The text also mentions avripiprazole fold change and a relative change from a reference value. However, the text contains some errors and incomplete phrases, making it difficult to interpret some of the information.*
Figure5 - Figure5
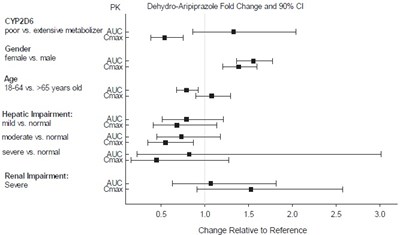
This seems to be a table depicting a study on the pharmacokinetics of Aripiprazole, a drug used to treat certain mental illnesses. The table compares how the drug is metabolized by individuals with different characteristics such as their CYP2D6 status (poor vs. extensive metabolizer), gender (female vs. male), and age (16-64 vs. >65 years old). Additionally, the table looks at how Aripiprazole is affected by hepatic impairment (mild vs. moderate vs. severe vs. normal) and renal impairment (severe). The last column appears to show the Dehydro-Aripiprazole Fold Change and 90% CI, which may indicate the effectiveness of the drug in each group.*
Figure 6 - figure 6

This appears to be a graph showing the proportion of subjects experiencing relapse over time for a drug called Ariplprazole compared to a placebo, with the number of subjects at risk shown below the graph. However, it is not clear what condition the drug is being tested for or any other specific details.*
Figure 7 - figure 7
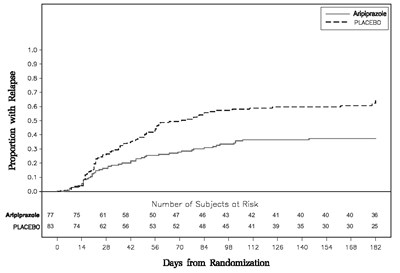
This text appears to be a table comprising medication names (Aripiprazole and Placebo), with proportions and the number of subjects at risk. The table also includes the days from randomization, with the number of subjects still under observation at each interval. There is no context provided or indication of what sort of study this data relates to.*
Figure 8 - figure 8

The text seems to be a table presenting the proportion of relapse for Aripiprazole and Placebo along with the number of subjects at risk for each treatment. The table also includes days from randomization. No further information is available.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.


