Product Images Donepezil Hydrochloride
View Photos of Packaging, Labels & Appearance
- fig-1 - donepezil figure 1
- fig-10 - donepezil figure 10
- fig-11 - donepezil figure 11
- fig-12 - donepezil figure 12
- fig-13 - donepezil figure 13
- fig-2 - donepezil figure 2
- fig-3 - donepezil figure 3
- fig-4 - donepezil figure 4
- fig-5 - donepezil figure 5
- fig-6 - donepezil figure 6
- fig-7 - donepezil figure 7
- fig-8 - donepezil figure 8
- fig-9 - donepezil figure 9
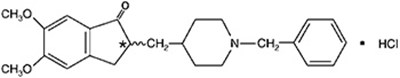
- str - donepezil str
- Label - lbl713352022
Product Label Images
The following 15 images provide visual information about the product associated with Donepezil Hydrochloride NDC 71335-2022 by Bryant Ranch Prepack, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
fig-10 - donepezil figure 10

This is a chart showing the cumulative percentage of patients in a clinical trial comparing the effects of Donepezil Hydrochloride 10mg/day versus placebo on the ADCS-ADL (Alzheimer's Disease Cooperative Study-Activities of Daily Living) change from baseline. The chart shows that there was a greater improvement in the ADCS-ADL in patients taking Donepezil Hydrochloride compared to those taking placebo.*
fig-11 - donepezil figure 11
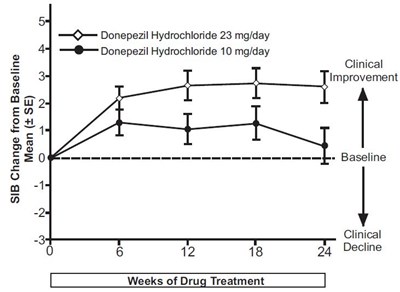
The text describes a chart showing the effects of two doses (23 mg/day and 10 mg/day) of Donepezil Hydrochloride on clinical improvement and decline over a period of 24 weeks. The chart is split into two sections measuring baseline and drug treatment. Not available information is provided about the context or purpose of the study.*
fig-12 - donepezil figure 12

This is a graph showing the cumulative percentage of patients who received either 23 mg/day of Donepezil Hydrochloride or a placebo. The x-axis shows the change from baseline in SIB (Severe Impairment Battery) score and the y-axis shows the percentage of patients.*
fig-13 - donepezil figure 13

This is a chart showing the percentage of patients who were given either 10mg or 23mg of Donepezil Hydrochloride per day and their corresponding CIBIC-plus ratings. The ratings range from markedly improved to markedly worse, with moderate and minimal improvements or worsenings in between.*
fig-2 - donepezil figure 2

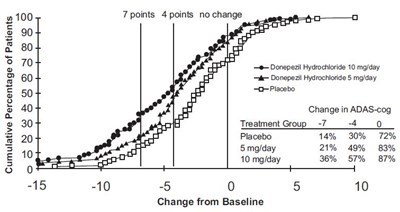
This is a comparison of the effects of Donepezil and Placebo on ADAS-cog (cognitive function) in patients. Donepezil is given in two doses, 5mg and 10mg Hydrochloride, and the placebo group does not receive any medication. The table shows the change in ADAS-cog scores from baseline for each group, -7 and -4 for the 5mg and 10mg Donepezil groups, respectively, and no change for the placebo group. The cumulative percentage of patients in each group who experienced a certain level of improvement in ADAS-cog from baseline is also shown.*
fig-3 - donepezil figure 3

This looks like a table that shows the percentage of patients taking Donepezil Hydrochloride medication at different doses (10mg/day and 5mg/day) and a placebo. The table also indicates the level of improvement in patients based on the CIBIC-plus Rating. The rating ranges from markedly improved to markedly worse.*
fig-5 - donepezil figure 5
This text represents a chart showing the cumulative percentage of patients and their corresponding change in ADAS-Cog (Alzheimer's Disease Assessment Scale - cognitive subscale) score in various treatment groups (Donepezil Hydrochloride 10 mg/day, Donepezil Hydrochloride 5 mg/day, and Placebo) over time. The chart also shows the change from baseline scores in each group.*
fig-7 - donepezil figure 7

This is a graph that shows the change from baseline in SIB (Severe Impairment Battery) score for patients who either received Donepezil Hydrochloride 10 mg/day or a placebo. The graph plots the mean (+ SE) scores over a period of 6 months. The x-axis represents the months of drug treatment, while the y-axis represents the SIB score. The graph indicates that clinical improvement was observed for patients who received Donepezil Hydrochloride compared to the placebo group.*
fig-8 - donepezil figure 8

The graph displays the cumulative percentage of patients who took Donepezil Hydrochloride 10mg/day and placebo, and their corresponding SIB (Severe Impairment Battery) change from baseline over 75 days. The x-axis shows the time in days, while the y-axis indicates the SIB change (in points). The line graph of the percentage of patients who took Donepezil Hydrochloride 10mg/day shows a higher SIB change from baseline than that of the placebo.*
Label - lbl713352022

This is a description of a medication known as Donepezil 5 milligrams. It comes in a white circular tablet form with the imprint ML;89. The text also contains instructions to keep the drug out of reach from children and store it at room temperature between 20-25°C. The medication is produced by Macleods Pharmaceutical Ltd. with a quantity of 90 tablets per bottle. The expiration date has not been mentioned. There is also a code NDC 7133520221 04524901523487.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.