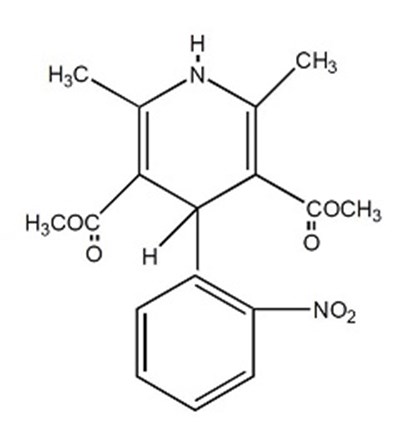
Product Images Nifedipine
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 4 images provide visual information about the product associated with Nifedipine NDC 71335-2132 by Bryant Ranch Prepack, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
02 - image 02
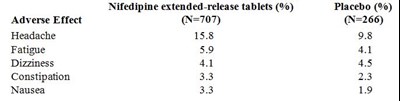
This is a list of adverse effects caused by Nifedipine extended-release tablets compared to placebo based on a study with 707 participants. The adverse effects include headache, fatigue, dizziness, constipation, and nausea, with percentages ranging from 3.3% to 33%. The placebo group had lower percentages of adverse effects.*
03 - image 03

This is a table containing the percentage of a number of adverse effects experienced by two groups of patients (one group taking Nifedipine capsules and the other group taking a placebo). The adverse effects listed include dizziness, lightheadedness, giddiness, flushing, heat sensation, headache, weakness, nausea, heartburn, muscle cramps, tremor, peripheral edema, nervousness, mood changes, palpitations, dyspnea, cough wheezing, nasal congestion, and sore throat.*
Label - lbl713352132

This seems to be a medication label with information about a 90mg tablet of Nifedipine ER, manufactured by Novast Laboratories. The label includes instructions to store the medication at room temperature and keep it out of reach of children, as well as a lot number and expiration date.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.