FDA Label for Cellbn First Care Cleanser
View Indications, Usage & Precautions
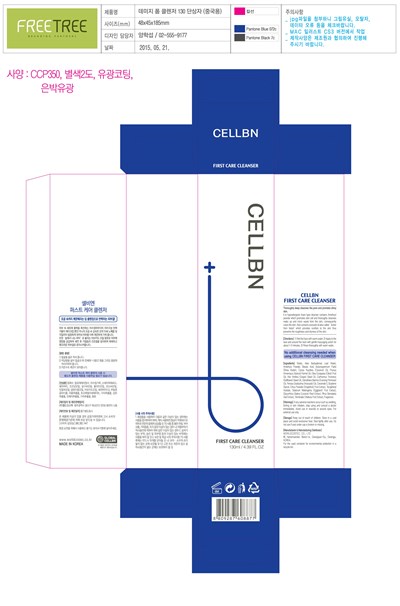
Cellbn First Care Cleanser Product Label
The following document was submitted to the FDA by the labeler of this product Worldcostec.co.,ltd. The document includes published materials associated whith this product with the essential scientific information about this product as well as other prescribing information. Product labels may durg indications and usage, generic names, contraindications, active ingredients, strength dosage, routes of administration, appearance, warnings, inactive ingredients, etc.
Otc - Active Ingredient
Stearic Acid
Inactive Ingredient
Water, Butylene Glycol, Etc
Otc - Purpose
Skin Protectant - Cleasing
Otc - Keep Out Of Reach Of Children
Keep out of reach of the children
Indications & Usage
- Wet the face with warm water.
- Apply to the face and around the neck with gently massaging action for abount 1~2 minutes.
- Rinse thoroughly with warm water.
Warnings
1. If the following symptoms occur after product use, stop using the product immediately and consult a dermatologist (continuous use can exacerbate the symptoms).
1) Occurrence of red spots, swelling, itchiness, and other skin irritation
2) If the symptoms above occur after the application area is exposed to direct sunlight
2. Do not use on open wounds, eczema, and other skin irritations
3. Precaution for Storage and Handling
1) Close the lid after use
2) Keep out of reach of infants and children
3) Do not to store in a place with high/low temperature and exposed to direct sunlight
4. Use as avoiding eye areas.
Dosage & Administration
for external use only
Package Label.Principal Display Panel
* Please review the disclaimer below.