Product Images Divalproex Sodium
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 7 images provide visual information about the product associated with Divalproex Sodium NDC 71610-032 by Aphena Pharma Solutions - Tennessee, Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Bottle Label 500mg - 71610 032 53

This is a bottle of Divalproex Sodium Delayed-Release tablets with a strength of 500mg and 60 tablets in the bottle. The brand name on the label appears to be "Ovalproex Sodum Delayed-Release", however, there seems to be a typo where the "d" is missing from "Sodium".*
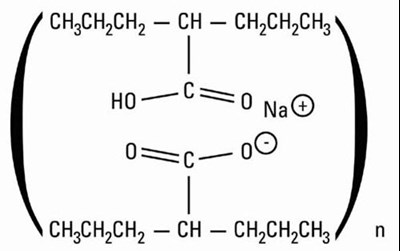
Figure 1 - divalproex 02

The text represents the results of two studies (Study 1 and Study 2) using the YMRS and MRS scales. The percentage values may indicate the level of severity of some conditions or symptoms measured by the scales. Other than that, no other information is available to determine the exact nature of the studies or their purpose.*
Figure 2 - divalproex 03

This appears to be a list of medication names, but there is no useful description that can be generated from it without additional context.*
Figure 3 - divalproex 04

This document appears to be a representation of a chart or graph showing the percentage reduction in CPS (compound per second) rate for high and low dose cases. The chart also shows the percentage of patients who experienced improvement, no change, or worsening. It is unclear what the data is describing and what the units are.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.