Product Images Lyrica
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 18 images provide visual information about the product associated with Lyrica NDC 71610-071 by Aphena Pharma Solutions - Tennessee, Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Bottle Label 25 mg - 71610 0071 30

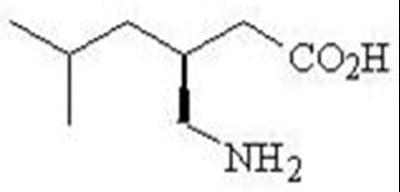
This is a prescription medication called Pregabalin, commonly sold under the brand name Lyrica. The pack contains 30 capsules of 25mg strength. The medication has a warning regarding the possibility of it being habit-forming. The rest of the text is not readable.*
Cockcroft and Gault equation - lyrica 01
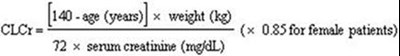
This appears to be a medical formula for calculating the estimated glomerular filtration rate (eGFR) of a patient. The formula takes into account the patient's weight, serum creatinine levels, and gender (by adjusting the value of Cl for female patients). However, there is a small error in the text, as the character "fi" is not recognized, and should be replaced with "fi".*
Figure 1 - lyrica 03

Percent of Patients Improved data is available but the rest of the text is not-readable.*
Figure 5 - lyrica 07

This appears to be a statistical report on the percentage of patients who showed improvement in pain from baseline after taking Pregabalin and Progabalin at various doses during the day. However, the information is incomplete and not presented in a clear manner, making it difficult to accurately interpret.*
Figure 8 - lyrica 10

The text seems to show a table displaying the responder rates in percentages. The first two columns appear to be empty, while the third column presents percentages ranging from 40.6% to 226%. The table includes a row indicating the treatment used, with two options being "Placebo 2.5 mg/kglday" and "10 mgrkgiday". It also shows values of 93% and 103% for each treatment, respectively.*
Figure 9 - lyrica 11

This text is not available as it contains incomplete sentences, symbols and unintelligible characters.*
Figure 11 - lyrica 13

This is a table that represents the percentage of subjects who improved between baseline and week 12 on Pregabalin medication. The percentages range from 50 to 100, with the majority being between 90 and 100.*
Logo - lyrica 14a

This text is a statement indicating the distributor of a product, Parke-Davis, which is a division of Pfizer Inc, located in NY, NY.*
Logo - lyrica 14b

This is a description of a pharmaceutical product indicating that it is distributed by Parke-Davis, a division of Pfizer Inc located in NY, NY 10017.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.