Product Images Losartan Potassium
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 7 images provide visual information about the product associated with Losartan Potassium NDC 71610-241 by Aphena Pharma Solutions - Tennessee, Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure1 - losartan fig1
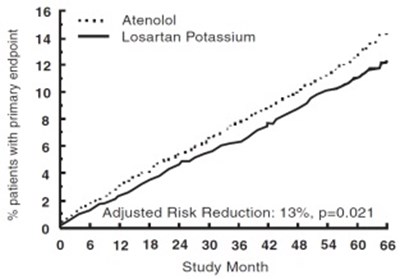
This text appears to be a medical or scientific study regarding the drugs Atenolol and Losartan Potassium. The only identifiable information is the "Adjusted Risk Reduction" of 13% and a chart or graph showing study months. There is not enough context to determine the purpose or results of the study.*
Figure-4 - losartan fig4
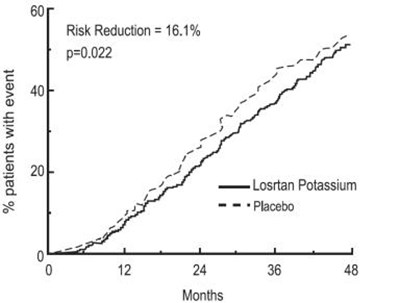
This appears to be a statistical analysis or report of a clinical trial involving a medication called "Losttan Potassium" and a placebo. The text mentions the percentage of patients who experienced an "event" (not specified), with 60% of patients on Losttan Potassium and 40% on placebo experiencing the event. The report also states a risk reduction of 16.1% and a p-value of 0.022, which may relate to a comparison of the incidence of the event between the two groups. The text does not provide further details, and it is unclear what the specific outcomes or implications of the trial may be.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.




