Product Images Divalproex Sodium
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 6 images provide visual information about the product associated with Divalproex Sodium NDC 71610-289 by Aphena Pharma Solutions - Tennessee, Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure 1 - divalproex fig1

The text describes a figure labeled "Figure 1" featuring the effects of Divalproex Sodium Delayed-Release Tablets compared to a placebo in reducing the CPS rate. The figure shows that the medication can result in a 50% improvement in CPS rate in patients.*
Figure 2 - divalproex fig2

The text corresponds to part of a figure showing the reduction in CPS rate for high and low dose Divalproex Sodium Delayed-Release Tablets. It indicates the percentage of patients who improved, had no change, or worsened. No further information is given.*
fig-3 - divalproex fig3
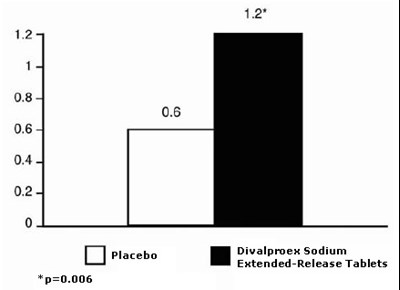
This is a list of numerical values followed by the name of a medication: Divalproex Sodium Extended-Release Tablets. No additional information or context is provided.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.


