Product Images Buspirone Hydrochloride
View Photos of Packaging, Labels & Appearance
- Structural Formula - 103f116c e4f4 47d7 82aa 59a2962e465e 01
- 15 mg (the entire tablet) - 103f116c e4f4 47d7 82aa 59a2962e465e 02
- 10 mg (two-thirds of a tablet) - 103f116c e4f4 47d7 82aa 59a2962e465e 03
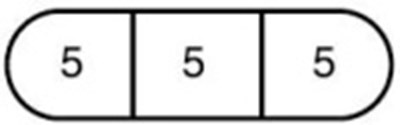
- 5 mg (one-third of a tablet) - 103f116c e4f4 47d7 82aa 59a2962e465e 04
- 7.5 mg (one-half of a tablet) - 103f116c e4f4 47d7 82aa 59a2962e465e 05
- 30 mg (the entire tablet) - 103f116c e4f4 47d7 82aa 59a2962e465e 06
- 20 mg (two-thirds of a tablet) - 103f116c e4f4 47d7 82aa 59a2962e465e 07
- 10 mg (one-third of a tablet) - 103f116c e4f4 47d7 82aa 59a2962e465e 08
- 15 mg (one-half of a tablet) - 103f116c e4f4 47d7 82aa 59a2962e465e 09
- Bottle Label 7.5 mg - 71610 0360 60
- Aphena Pharma Solutions - TN - Aphena
- Breaking Multi-Scored Tablet - image 01
Product Label Images
The following 12 images provide visual information about the product associated with Buspirone Hydrochloride NDC 71610-360 by Aphena Pharma Solutions - Tennessee, Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.