Product Images Pregabalin
View Photos of Packaging, Labels & Appearance
- Bottle Label 50 mg - 71610 0350 30
- Bottle Label 300 mg - 71610 0351 30
- Bottle Label 150 mg - 71610 0352 53
- Bottle Label 75 mg - 71610 0356 53
- Bottle Label 100 mg - 71610 0357 53
- Bottle Label 225 mg - 71610 0358 53
- Bottle Label 200 mg - 71610 0361 53
- Bottle Label 25 mg - 71610 0365 53
- Aphena Pharma Solutions - TN - Aphena
- Figure - pregabalin 01
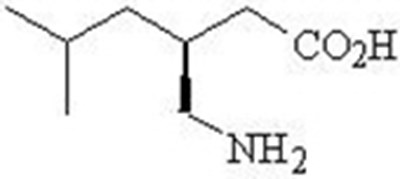
- Chemical Structure - pregabalin 02
- Figure 1 - pregabalin 03
- Figure 2 - pregabalin 04
- Figure 3 - pregabalin 05
- Figure 4 - pregabalin 06
- Figure 5 - pregabalin 07
- Figure 6 - pregabalin 08
- Figure 7 - pregabalin 09
- Figure 8 - pregabalin 10
- Figure 9 - pregabalin 11
- Figure 10 - pregabalin 12
- Figure 11 - pregabalin 13
- Figure 12 - pregabalin 14
- Logo - pregabalin 15
- Logo - pregabalin 15a
Product Label Images
The following 25 images provide visual information about the product associated with Pregabalin NDC 71610-365 by Aphena Pharma Solutions - Tennessee, Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Bottle Label 25 mg - 71610 0365 53

This is a description of a medication called Pregabatin with a Nock number of 71610-0365-53. There are some illegible characters, but no useful information is available.*
Figure - pregabalin 01

The text contains laboratory results for serum creatinine, specified for female patients. It includes the numeric values of 1050 and 72. No further information is available.*
Figure 2 - pregabalin 04

The table displays the percentage of patients who showed improvement in pain after taking Pregabalin 106 mg three times per day. There are three values in the first column representing the percentage of patients improved - 100%, 20% and 10%. The second column reads "Pregabalin 106 mg three times a day". The rest of the text is not-readable, appearing to be scrambled output.*
Figure 3 - pregabalin 05

The text shows a chart representing the percentage of improvement in pain from a baseline for different doses of Pregabalin and Placebo. However, specific details about the chart's context are not available.*
Figure 4 - pregabalin 06

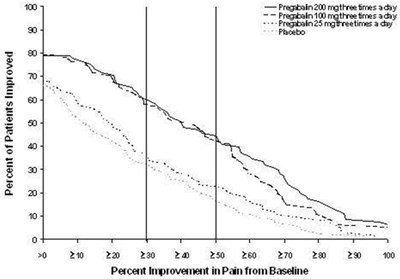
This appears to be a chart showing the percent of patients who improved with different treatments (Pregabalin 200 mg, Pregabalin 100 mg, and Placebo) and the percent improvement in pain from baseline. Unfortunately, there seems to be some missing or unreadable characters, making the chart difficult to interpret properly.*
Figure 5 - pregabalin 07

This is a graph showing the percentage of patients' improvement in pain from the baseline. The X-axis represents the percentage of improvement, and the Y-axis represents different treatments: Pregabalin 100 mg three times a day, Pregabalin 60 mg three times a day, and Placeno. However, the graph seems to be corrupted and has some random characters that make it difficult to interpret.*
Figure 8 - pregabalin 10

This appears to be a table displaying the responder rate (%) for different treatments. The table shows three different responder rates corresponding to placebo, 2.5 mg/kg/day, and 10 mg/kg/day treatments. The values for placebo, 2.5 mg/kg/day, and 10 mg/kg/day are 40.6%, 29.1%, and 22.6%, respectively. The table also lists the number of responders for each treatment, with 93 responders for placebo treatment, 103 responders for the 2.5 mg/kg/day treatment, and 96 responders for the 10 mg/kg/day treatment.*
Figure 9 - pregabalin 11

This is a chart showing the percentage of patients who have experienced improvement in pain from baseline using different doses of Pregabalin. The chart includes a daily dose of 600mg, 450mg, and 300mg. The placebo is also shown in the chart. The percentages range from 2% to 90%.*
Figure 10 - pregabalin 12

This appears to be a graph showing the estimated percentage of subjects without LTR (likely to respond) in a study involving a placebo and Pregabalin over a period of 180 days. The x-axis indicates the number of days while the y-axis shows the percentage of subjects without LTR. The graph shows that there is a higher percentage of subjects without LTR in the Pregabalin group compared to the placebo group.*
Figure 11 - pregabalin 13

This is a table that shows the percentage of subjects improved on Pregabalin and Placebo over 12 weeks. The table also displays the number of subjects that improved.*
Logo - pregabalin 15

This is a text regarding the Greenstone brand. It is distributed by Greenstone LLC located at Peapack, NJ 07977.*
Logo - pregabalin 15a

This is a description of Greenstone LLC, a distributor of a brand called GREENSTONE® located in Peapack, NJ 07977.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.