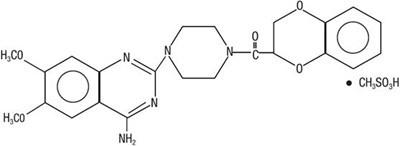
Product Images Doxazosin
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 5 images provide visual information about the product associated with Doxazosin NDC 71610-482 by Aphena Pharma Solutions - Tennessee, Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Table 3 - doxazosin 02

The text represents tables of results from clinical studies evaluating the efficacy of doxazosin in treating benign prostatic hyperplasia (BPH). The studies measured changes in maximum flow rate and symptom score of patients before and after taking placebo or doxazosin at various doses. Study 1 involved titration to the maximum dose of 8 mg, while Study 2 involved titration to a fixed dose over 14 weeks. Study 3 involved titration to a fixed dose over 12 weeks. The results show higher mean maximum flow rates and symptom score improvements in patients taking doxazosin compared to placebo, with some statistically significant differences.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.