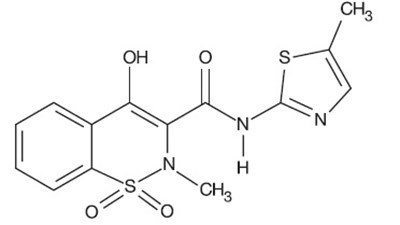
Product Images Meloxicam
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 9 images provide visual information about the product associated with Meloxicam NDC 71610-583 by Aphena Pharma Solutions - Tennessee, Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Adverse Reactions - Adverse Reactions
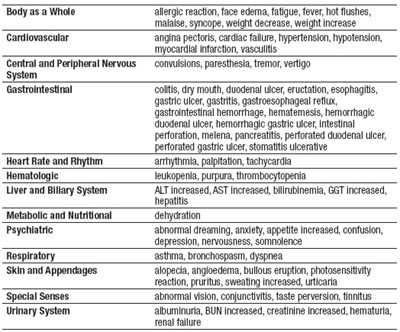
This text appears to be a list of various side effects or symptoms that may be experienced in relation to different bodily systems, such as cardiovascular, respiratory, gastrointestinal, and urinary. Some examples include allergic reactions, weight changes, hypertension, vertigo, gastric ulcers, abnormal heart rate, anemia, liver issues, and urinary problems. It also mentions psychiatric symptoms such as depression and somnolence. The text may be a compilation of potential adverse effects of a particular medication or health condition.*
Table 1a - Table 1a
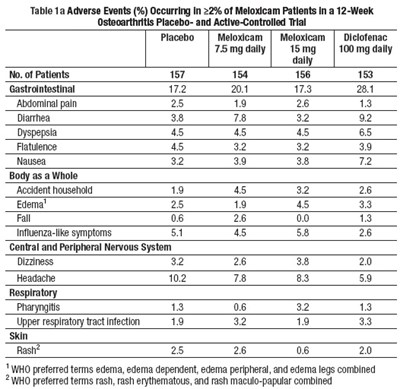
The text describes adverse events occurring in patients taking different doses of meloxicam and diclofenac in a 12-week osteoarthritis trial. The table provides a breakdown of the percentage of patients experiencing adverse events for each medication, including gastrointestinal problems, body-wide effects, nervous system effects, and skin issues. The table reveals that meloxicam at 15mg and 100mg daily reported the highest number of adverse events in most categories. The text provides aggregated results based on WHO-preferred terms for some adverse events.*
Table 1b - Table 1b
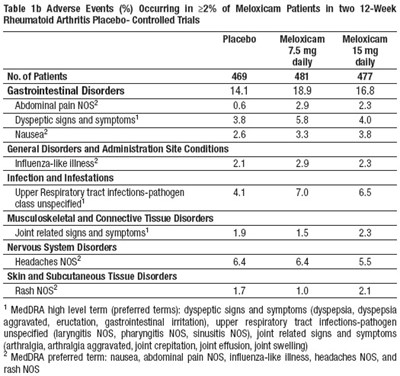
The table shows the adverse events (%) experienced by more than 2% of Meloxicam patients in two 12-week placebo-controlled trials conducted for Rheumatoid Arthritis. The Meloxicam patients were divided into two sub-groups - one received 7.5 mg/day and the other 15 mg/day. The adverse events are classified as per the body system affected. The adverse events included gastrointestinal disorders, infections and infestations, musculoskeletal and connective tissue disorders, and nervous system disorders, among others. The most common symptoms were dyspepsia, nausea, joint pain, headache, and rash.*
Table 4 - Table 4
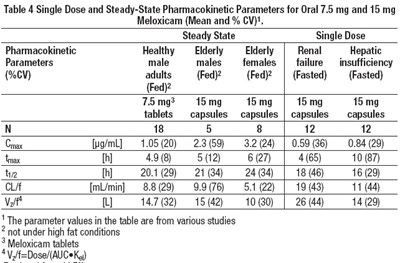
This is a table that presents pharmacokinetic parameters for oral 7.5mg and 15mg Meloxicam, including steady-state and single dose pharmacokinetics in healthy adults and elderly males and females without renal or hepatic insufficiency, but also in elderly and renal or hepatic insufficiency patients. The table includes multiple parameter values obtained from various studies. The studies were not conducted under high fat conditions. The Meloxicam administered was in the form of tablets and capsules.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.