Product Images Pregabalin
View Photos of Packaging, Labels & Appearance
- Bottle Label 75 mg - 71610 0758 30
- Bottle Label 200 mg - 71610 0759 30
- Bottle Label 225 mg - 71610 0760 46
- Aphena Pharma Solutions - TN - Aphena
- equation - pregabain gault equation
- fig-01 - pregabalin figure 01
- fig-02 - pregabalin figure 02
- fig-03 - pregabalin figure 03
- fig-04 - pregabalin figure 04
- fig-05 - pregabalin figure 05
- fig-06 - pregabalin figure 06
- fig-07 - pregabalin figure 07
- fig-09 - pregabalin figure 09
- fig-10 - pregabalin figure 10
- fig-11 - pregabalin figure 11
- fig-12 - pregabalin figure 12
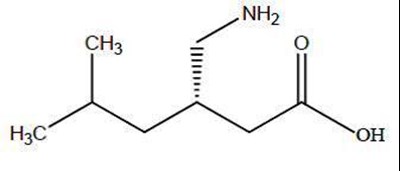
- str - pregabalin structure
Product Label Images
The following 17 images provide visual information about the product associated with Pregabalin NDC 71610-758 by Aphena Pharma Solutions - Tennessee, Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
equation - pregabain gault equation
This text appears to be a formula for calculating CLCr (Creatinine Clearance) based on age, weight, and serum creatinine levels. The formula suggests that for male patients, CLCr is equal to 1140 minus the age in years multiplied by weight in kilograms. For female patients, CLCr should be multiplied by 0.85. It is important to note that the quality of the text obtained from is not perfect, so some of the information may be misinterpreted or missing.*
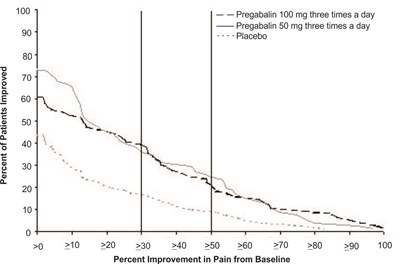
fig-01 - pregabalin figure 01

Percent of Patients Improved: This text presents data on the percentage of patients who experienced improvement from baseline in pain levels. Unfortunately, the specific details of the improvement and the source from which the data was obtained are not provided, making it difficult to evaluate the significance of the information.*
fig-02 - pregabalin figure 02

This text provides information about the percent improvement in pain from baseline for patients taking Pregabalin 100 mg three times a day versus a placebo. It shows the percentages of patients improved at different levels, ranging from 10% to 100%.*
fig-03 - pregabalin figure 03
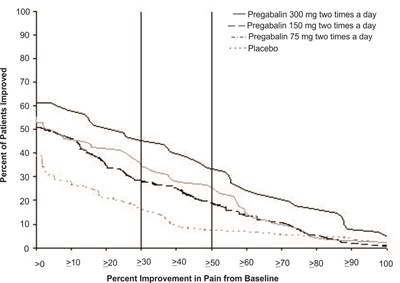
This text provides information on the percent of patients improved with different doses of Pregabalin throughout the day, as well as a placebo. The text also includes a chart showing the percent improvement in pain from baseline.*
fig-04 - pregabalin figure 04

The text provided contains a chart showing the percentage of patients who experienced improvement in pain while taking different doses of pregabalin three times a day, as well as a placebo. The chart also indicates the percent improvement in pain from the baseline for various levels.*
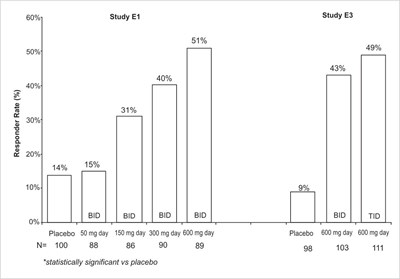
fig-09 - pregabalin figure 09

This is a table showing the percent of patients who improved based on different doses of pregabalin (600 mg, 450 mg, and 300 mg) compared to a placebo. The table also includes different levels of pain improvement from baseline (greater than 20%, 30%, 40%, etc.).*
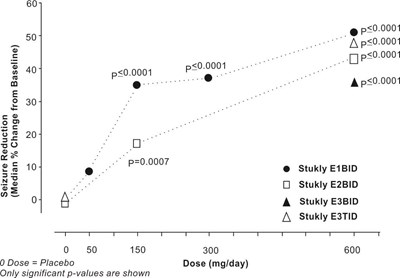
fig-10 - pregabalin figure 10

This text appears to be a table showing the estimated percentage of subjects without low income rate (LIR) for different values ranging from 0% to 100%. It also mentions the use of pregabalin and placebo, and includes a scale of days ranging from 80 to 180.*
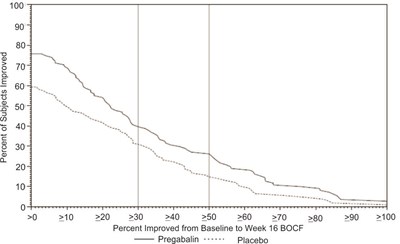
fig-11 - pregabalin figure 11
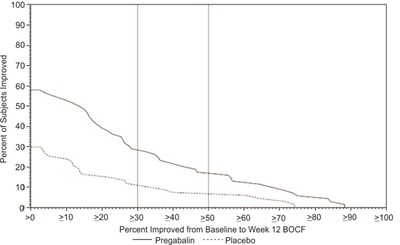
Percent of Subjects Improved: This data represents the percentage of subjects who showed improvement from baseline to week 12 according to the BOCF (Baseline Observation Carried Forward) method. The graph shows the percent improvement on the y-axis and the range of improvement levels on the x-axis, ranging from 0 to 100. The study compares the effectiveness of Pregabalin versus a placebo, although further details about the specific outcomes are not available.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.