Product Images Nifedipine
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 8 images provide visual information about the product associated with Nifedipine NDC 71610-841 by Aphena Pharma Solutions - Tennessee, Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Label - 71610 0841 60

This text provides information about a medication called Nifedipine ER, USP in the form of 30mg tablets. The packaging contains 90 tablets and is manufactured by Novast Laboratories Ltd. in Nantong, China, and packaged by Aphena Pharma in Cookeville, Tennessee. The NDC number for this product is 71610-841-60 and the GTIN is 1037110841602. The batch number is 1 and the serial number is P022485701. It is important to keep this medication and all drugs out of the reach of children.*
image2 - image 02
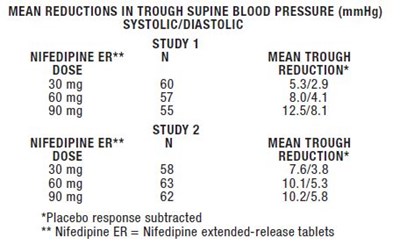
This text provides information on mean reductions in trough supine blood pressure (in mmHg) for systolic and diastolic readings with different doses of Nifedipine extended-release tablets from studies 1 and 2. The data shows how the blood pressure readings changed with the different doses of Nifedipine ER compared to placebo.*
image3 - image 03

This table provides data on the percentage of adverse effects experienced by patients taking nifedipine extended-release tablets compared to those taking placebo. Common adverse effects include headache, flushing/heat sensation, dizziness, fatigue/asthenia, nausea, and constipation. The study involved 370 patients in the nifedipine group and 126 patients in the placebo group.*
image4 - image 04

These are tablets of different strengths with color markings indicating the dosage. The tablets have either 30 mg, 60 mg, or 90 mg of active ingredient. They are marked with the Novast logo on one side and numbers 33, 36, or 39 on the other side to identify their respective strengths.*
image5 - image 05

This is a list detailing different bottle sizes and strengths of a medication identified by its NDC codes. The medication comes in bottles of 100 tablets with strengths of 30 mg, 60 mg, and 90 mg. The NDC codes for each are 50742-620-01, 50742-621-01, and 50742-622-01 respectively. These codes are used to uniquely identify the medication and dosage form for billing and tracking purposes.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.


