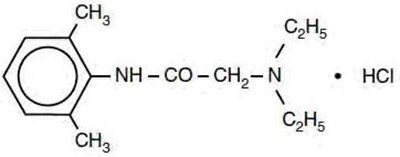Product Images Lidocaine Hydrochloride
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 3 images provide visual information about the product associated with Lidocaine Hydrochloride NDC 71872-7256 by Medical Purchasing Solutions, Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
71872-7256-01.PDP - 71872 7256 01

2% Lidocaine HCI Injection is a sterile, non-pyrogenic solution in a multiple-dose vial with Lidocaine hydrochloride equivalent to 17.8 mg Lidocaine per mL. It also contains sodium chloride, methylparaben and hydrochloric acid. The injection is intended for infiltration and nerve block, but not for caudal or epidural use. It can be resterilized by autoclaving and should not be used if discolored or contains a precipitate. The usual dosage and prescribing information are available in the package insert. The vial stopper doesn't contain natural rubber latex. The product is distributed by AuroMedics Pharma LLC and repackaged by Medical Purchasing Solutions.*
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 2% 1,000 mg per 50 mL (20 mg / mL) - 50 mL Container Label - lidocaine fig11

This is a description for a medication with the NDC code 55150-256-50 used for infiltration and nerve block. It is a 50 mL multiple dose vial of lidocaine hydrochloride injection, USP, sterilized and nonpyrogenic. The preservative used in the medication is Methylparaben. The medication should not be used if the solution is discolored or has a precipitate. It is recommended to store the medication at 20°C to 25°C (68°F to 77°F) and is not suitable for caudal or epidural use. The medication is distributed by AuroMedics Pharma LLC and made in India.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.