Product Images Venlafaxine Hydrochloride
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 13 images provide visual information about the product associated with Venlafaxine Hydrochloride NDC 71921-173 by Florida Pharmaceutical Products, Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Venlafaxine-01 - ff9008be 0609 415c 810f c2bbbbdf305e 01
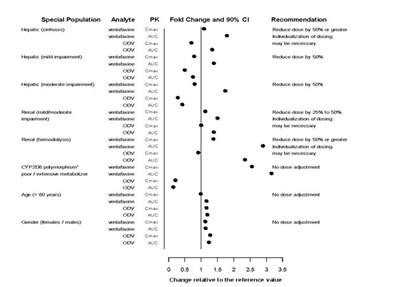
This appears to be a table with some medical information related to interacting drugs, analytes, and PK fold change, but it is not clear and contains errors, so it is difficult to generate a precise description of what it represents.*
Venlafaxine-02 - ff9008be 0609 415c 810f c2bbbbdf305e 02

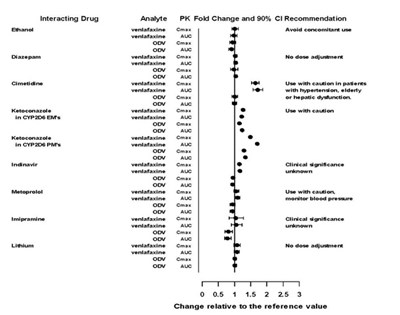
This appears to be a table with information about an interacting drug called Matoprotol, and its effect on a specific analyte. However, parts of the text are not readable and therefore, it is not possible to provide a useful description.*
ff9008be-0609-415c-810f-c2bbbbdf305e-04 - ff9008be 0609 415c 810f c2bbbbdf305e 04
This is a partial text and does not provide enough information to generate a useful description. It appears to be a mix of random characters and one drug name "venlafaxine hydrochloride" which is used to treat depression and anxiety disorders.*
ff9008be-0609-415c-810f-c2bbbbdf305e-05 - ff9008be 0609 415c 810f c2bbbbdf305e 05

This is a description of a medication container for venlafaxine HCI extended-release capsules, USP, with a strength of 37.5mg. It contains 30 capsules and comes with a medication guide that should be provided to each patient. The usual dosage is provided in the accompanying prescribing information. The package is intended to be dispensed as a unit and is sealed for protection. It was manufactured by Inventia Healthcare Limited, located in India, for Florida Pharmaceutical Products LLC. The container has a 2D barcode and an unvarnished area which measures 45 x 15 mm.*
ff9008be-0609-415c-810f-c2bbbbdf305e-06 - ff9008be 0609 415c 810f c2bbbbdf305e 06
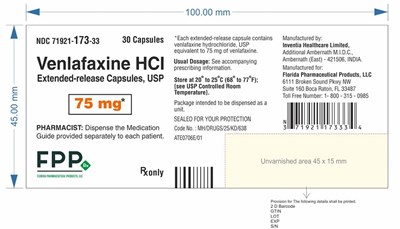
This is a description of a medication containing 30 capsules of extended-release Venlafaxine HCI, USP at 75mg. The medication guide provided for each patient should be dispensed by the pharmacist. The usual dosage is unknown and the medication should be stored at 20° to 25°C. The package is sealed for protection and should be dispensed as a unit. The manufacturer is Inventia Healthcare Limited in India and produced for Florida Pharmaceutical Products, LLC in the USA. The code for the medication is 71921-173-33 and contains a 2D barcode. Additionally, there is an unvarnished area that measures 45 x 15 mm.*
ff9008be-0609-415c-810f-c2bbbbdf305e-07 - ff9008be 0609 415c 810f c2bbbbdf305e 07

This appears to be packaging information for capsules of Venlafaxine HCI, containing 150mg of the active ingredient, which is venlafaxine hydrochloride. The package contains 30 extended-release capsules, which should be stored at room temperature. The manufacturer is Invenia Healthcare Limited, and the capsules are to be dispensed with an accompanying medication guide. There also seems to be a barcode and lot number included for tracking purposes.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.