Product Images Wakix
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 11 images provide visual information about the product associated with Wakix NDC 72028-178 by Harmony Biosciences, Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure 1 - wakix 02

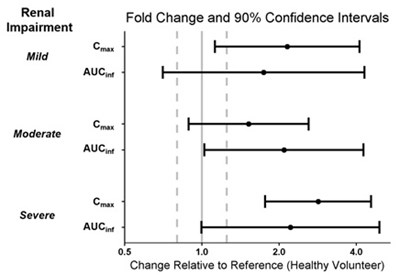
This appears to be a table showing the impact of hepatic impairment (mild and moderate) on the AUC (area under the concentration-time curve) and fold change, along with their 90% confidence intervals, in relation to a reference healthy volunteer. The term "Crax" is not clear in this context and may be a misinterpretation of another word.*
Figure 3 - wakix 04

This text appears to show statistical information about "Fold Change and 90% Confidence Intervals" related to "Relative change to the reference (Extensive Metabolizers)". However, the text is incomplete and therefore it is not possible to provide a useful description.*
Figure 4 - wakix 05

This text appears to be a list of medications classified according to their inhibitory or inducing effects on certain enzymes in the body, such as CYP2D6 and CYP3A. It also mentions fold change and confidence intervals related to these medications, possibly indicating their potency or efficacy. Some of the listed medications include paroxetine, iraconazole, ritampicin, modatini, and sodium oxybate.*
4.45 mg Bottle Label - wakix 08

This is a description of the medication Wakix in tablet form, containing either 4.45mg of pitolisantas or 5mg of pitolisant hydrochloride. The dosage and administration instructions can be found in the package insert. It is to be stored at room temperature between 20°C to 25°C. It is manufactured for Harmony Biosciences, LLC. The given NDC number is 72028-045-03 with a product number of 70014523.*
4.45 mg Carton Label - wakix 09

WAKIX is a medication that comes in a container of 30 tablets and is made in France. Each tablet contains 4.45 mg of pitolisant hydrochloride. It is manufactured for Harmony Biosciences, LLC based in Plymouth Meeting, PA. The Harmony Biosciences name and logo are registered trademarks.*
17.8 mg Bottle Label - wakix 10

This is a description of a medication identified by the National Drug Code (NDC) 72028-178-03. The medication consists of tablets that contain 17.8 mg of pitolisantas and 20 mg of pitolisant hydrochloride, produced by Harmony Biosciences, LLC. The dosage and administration details are available in the package insert. The medication needs to be stored in a temperature range of 20°C to 25°C (68°F to 77°F).*
17.8 mg Carton Label - wakix 11

Wakix is a brand of pitolisant tablets that contain 17.8 mg of pitolisant hydrochloride. The dosage instructions for pitolisant are unknown, but users should see the package insert for this information. Every package contains 30 tablets, and the tablets should be stored at a temperature between 20°C to 25°C (68°F to 77°F). Harmony Biosciences, LLC manufactures Pitolisant tablets for Bioprojet Europe, Ltd. Each tablet container has a unique component code (70015547) that may be used for serialization and Datamatrix coding purposes.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.